| Monthly Tech-Tip | No tracking! No ads! No paywall! |
Click here for case-studies of Insight-Live fixing problems
You will see examples of replacing unavailable materials (especially frits), fixing various issues (e.g. running, crazing, settling), making them melt more, adjusting matteness, etc. Insight-Live has an extensive help system (the round blue icon on the left) that also deals with fixing real-world problems and understanding glazes and clay bodies.
Related Pictures
What can you do using glaze chemistry? More than you think!

This picture has its own page with more detail, click here to see it.
There is a direct relationship between the way ceramic glazes fire and their chemistry. These green panels in my Insight-live account compare two glaze recipes: A glossy and matte. Grasping their simple chemistry mechanisms is a first step to getting control of your glazes. To fixing problems like crazing, blistering, pinholing, settling, gelling, clouding, leaching, crawling, marking, scratching, powdering. To substituting frits or incorporating available, better or cheaper materials while maintaining the same chemistry. To adjusting melting temperature, gloss, surface character, color. And identifying weaknesses in glazes to avoid problems. And to creating and optimizing base glazes to work with difficult colors or stains and for special effects dependent on opacification, crystallization or variegation. And even to creating glazes from scratch and using your own native materials in the highest possible percentage.
Is that recipe you found online really what you think it is?

This picture has its own page with more detail, click here to see it.
It contains significant Frit P-25, I googled that to Digitalfire, it contains 1.8% fluorine! When much of that gases off during firing are you really equipped to deal with fluorine gas in your studio? One answer is to substitute my own frits (which of course do not contain fluorine). I did the calculations in my account at insight-live.com (see the recipes side-by-side). In the formula, I substituted the F (fluorine) for a mix of CaO/MgO. In the recipe, I substituted Ferro Frit 3110 (it is also a high-sodium frit) for the P-25, then I juggled the rest of the recipe to match up the oxides. More Gerstley Borate supplies the lost B2O3, more ball clay supplies the lost Al2O3 and less silica is needed (because the ball clay brought along more). I ended up with a recipe that is going to suspend much better (more clay) and was able to eliminate the whiting (because the rest of the new recipe is supplying more CaO). This recipe has a high thermal expansion, that means crazing. Would it not be easier to simply add a red stain to a clear glaze you already have that is working well on your clay bodies?
Cone 6 transparent way better without Gerstley Borate.
I surgically removed it to create G2926B!

This picture has its own page with more detail, click here to see it.
These are the original cone 6 Perkins Studio Clear (left) beside our fritted version (right). You cannot just substitute a frit for Gerstley Borate (GB), they have very different chemistries. But, using the calculation tools in my account at insight-live.com, I compensated for the differences by juggling other materials in the recipe. I even upped the Al2O3 and SiO2 a little on the belief they would dissolve in the more active melt the frit would create. I was right - a melt-flow GLFL test comparison (inset left) shows that the GB version flows less. Using this on ware exhibited another issue (after doing a IWCT test): Crazing. The very good melt flow on my G2926A fritted version is thus good news: It can accept more silica - the more silica, the more durable and craze resistant it will be. How much did it take? 10% more! That ultimately became the recipe for our standard G2926B cone 6 transparent.
Step-by-step to do a formula-to-batch in Insight-Live.com

This picture has its own page with more detail, click here to see it.
Insight-live does not automate formula-to-batch calculations, but it does assist in doing them. And it provides the tools to create an audit trail of test results, pictures and notes and a path to document subsequent adjustments. Along the way, you gain material knowledge and intuition. In this example, we derive the recipe of materials needed to source the oxide formula of a zinc clear cone 6 glaze (sourcing the oxides needed using a Ferro frit and other common raw materials). We'll create the target in a panel, start the batch in a panel beside it, supply the B2O3 from a frit and then the fluxes from feldspar, zinc and whiting. Then finish by rounding out the Al2O3 and SiO2 from kaolin and silica. The picture below shows the panels, the original target formula on the left and the final derived recipe on the right. The derived transparent glaze is on the inside of the mug and the outside is G3875, another zinc clear with iron and chrome added to produce the orange.
A large glaze batch mixing error rescued using glaze chemistry

This picture has its own page with more detail, click here to see it.
The person used Frit 3134 instead of 3124, it makes up 70% of the recipe. The glaze melted much more and ran off the ware. That sounds like an impossible-to-fix problem. It just so happens that these frits have very similar chemistry except for one thing: 3134 has almost no Al2O3. That means that kaolin can be added to the bad batch to source the missing Al2O3 (and replenish the shortage of SiO2 at the same time). Extra silica is also needed to restore the full SiO2. The new chemistry is not an exact match, the B2O3 is a little higher, but this should not be an issue. Of course a raw:calcine mix of kaolin is needed (to prevent the glaze from shrinking too much on drying and therefore cracking). From this calculation we can see that for every 100 grams of the original powder we need to add 10 EPK, 15 calcined kaolin and 17 silica. Of course, one would need to know the water content of the slurry, that is calculated as (weight wet - dry weight)/wet weight * 100. If the slurry was 50% water, for example, then every 200 grams of slurry would contain 100 grams of powder.
Substitute Ferro Frit 3134, using glaze chemistry, in three glaze types

This picture has its own page with more detail, click here to see it.
Can't get frit 3134 for glaze recipes? Can you replace it with frit 3124? No, 3124 has five times the amount of Al2O3 (the second most important oxide in glazes) and half the amount of B2O3 (the main melter). This ten-minute video presents a glaze chemistry approach that is easier to do than you probably think. It deals with three different glaze recipe types lacking sufficient clay to suspend the slurry. Learn to source the needed oxides from two other Ferro frits, 3110 (or Fusion F-75) and 3195 (Fusion F-2), and end up with at least 15% kaolin in each. A unique approach is required in each situation. Two of the calculations produce improved slurry properties and one yields a recipe of significantly lower cost. If you have a recipe that needs this and need help please contact us.
How I calculated a feldspar-to-frit replacement in a cone 10R clear glaze

This picture has its own page with more detail, click here to see it.
A screen shot of side-by-side panels in my account at Insight-live.com. On the left is the original G1947U recipe. On the right I have substituted frit 3110 as a higher-concentration-source of KNaO. This enabled actually increasing KNaO to get a better gloss and melt. I have introduced calcined kaolin (to ratio with the raw kaolin to control slurry and drying properties). I added frit 3249 to introduce low-expansion MgO to counterbalance the higher levels of KNaO. That frit, like the 3110, will not only melt things better simply because it itself was pre-melted, but it also brings along a little boron. That supercharges melting and enables an enhancement: The addition of more SiO2. The low thermal expansions of MgO, SiO2 and B2O3 counterbalance the increase that will occur as a result of the higher KNaO (0.15-0.26). Sound boring? When you see the unexpected results you might think differently (see the linked post below). I never considered using frits at cone 10R before, this success led to an improvement in my main silky matte glaze also.
Why do these cone 04 and 6 clear glazes have so similar a chemistry?

This picture has its own page with more detail, click here to see it.
The glaze on the left (as shown in my account at insight-live.com) is a crystal clear at cone 04. The high frit content minimizes micro-bubbles. The high B2O3 melts it very well (this has 0.66 B2O3, that is three times as high as a typical cone 6 glaze). The recipe on the right is the product of a project to develop a low-thermal-expansion fluid-melt transparent for cone 6 (with added colorants fluid melts produce brilliant and even metallic results and they variegate well). While the balance of fluxes (the red numbers in the formula) is pretty different, look how similar the B2O3, Al2O3 and SiO2 levels are (yellow, red and blue backgrounded numbers in the formula), these mainly determine the melting range. That means that a fluid-melt cone 6 glaze may actually be just a low temperature glaze being overfired to cone 6.
Why would a low fire transparent require four frits?

This picture has its own page with more detail, click here to see it.
To get the needed chemistry to avoid boron blue clouding (calcium borate crystals). The one on the right clouds, the other does not. Why? Differences in the chemistry (as seen in my account at insight-live.com). G2931K, on the left, has greater Al2O3 (which impedes the growth of crystals), lower CaO (starves their growth) and more boron (for better melting). There is actually no practical way to adjust the recipe on the right (by supplying MgO with talc and fiddling with frit percentages) to achieve this. Frit 3124 lacks Na2O and B2O3. 3134 has excessive CaO and almost zero Al2O3. Talc does not melt well enough. But Frit 3249 supplies the needed MgO and has lots of B2O3 and low CaO. And Frit 3110 has low CaO and supplies the needed Na2O.
Cannot get Nepheline Syenite? Here is what to do.

This picture has its own page with more detail, click here to see it.
Nepheline Syenite is similar to a feldspar. I have them open side-by-side here in my Insight-live.com account (the blue panels). In the "Analysis" column notice that Minspar has 9% more SiO2 and 5% less Al2O3. Minspar has 12% fluxes and Nepheline has more than 15%. If a recipe does not contain a significant percentage of Nepheline these differences might be tolerated but what if there is 30% or more? There is no combination of materials that has the chemistry of Nepheline (there is no way to take 10% of the SiO2 out of this feldspar, for example). But it is possible to take SiO2 out of a glaze containing Nepheline Syenite. Notice the two green recipe panels below: The changes made to the one on the right harmonize the oxide chemistry with the original on the left. Those changes were significant: 15% more feldspar, 12% less silica and 2% less kaolin. Notice how easy this was using a little chemistry!
Calculating a substitute for Minspar

This picture has its own page with more detail, click here to see it.
Why do this? We did not have it in stock and customers needed to mix recipes. When the chemistries of the two feldspars are very similar substitution is often not a problem, especially when a recipe only calls for 5 or 10%. However, when a recipe calls for a significant percentage the situation becomes much trickier (in our cone 6 test recipe, "Perfect Clear", 40% Minspar is needed). Feldspars are almost a glaze in themselves, just needing silica and alumina to shift their chemistry toward 'glazedom'. In this project I calculated a mix of materials, in my Insight-live.com account, that sources the same chemistry as Minspar. I made cone 6 GLFL tests comparing the pure Minspar and Minspar substitute (left) and comparing the Perfect Clear glaze with each feldspar (right). As you can see, the similarity in melt flow is stunning! This is a good demonstration of just how practical and valuable glaze chemistry calculation can be.
Does adding boron alone always increase glaze melt?

This picture has its own page with more detail, click here to see it.
Boron (B2O3) is like silica, but it is also a flux. Frits and Gerstley Borate supply it to glazes. In this test, I increased the amount of boron from 0.33 to 0.40 (using the chemistry tools in my insight-live.com account). I was sure that this would make the glaze melt more and have less of a tendency to craze. But as these GBMF tests for melt flow (10 gram GBMF test balls melted on porcelain tiles) show, that did not happen. Why? I am guessing that to get the effect B2O3 has to be substituted, molecule for molecule for SiO2 (not just added to the glaze).
Ravenscrag Plum Red recalculated to use frit instead of ulexite

This picture has its own page with more detail, click here to see it.
The original recipe, from which Ravenscrag Plum Red was derived, employed Gerstley Borate to source the boron (the melter). That was a problem because it gels glaze slurries (there is already 15% iron oxide present and that gels also). Thus ulexite was instead to source the boron. That was many year ago but now, ulexite is almost impossible for potters to get. We reformulated again, this time sourcing the boron from a frit (actually two frits). Frit 3249 was used to source some of the MgO needed (this glaze has high MgO levels) since it melts so well and also sources lots of boron. There is now a little less Ravenscrag Slip to suspend the slurry, but that is not a problem, the iron will gel it.
Comparing glaze melt fluidity balls with their chemistries

This picture has its own page with more detail, click here to see it.
Ten-gram GBMF test balls of these three glazes were fired to cone 6 on porcelain tiles. Notice the difference in the degree of melt? Why? You could just say glaze 2 has more frit and feldspar. There is a better explanation, compare these yellow and blue numbers: Glaze 2 and 3 have much more B2O3 (boron, the key flux for cone 6 glazes) and lower SiO2 (silica, it is refractory). But notice that glaze 2 and 3 have the same chemistry, but 3 is melting more? Why? Because of the mineralogy of Gerstley Borate. It release its boron earlier in the firing, getting the melting started sooner. Notice it also stains the glaze amber, it is not as clean as the frit. Notice the calculated thermal expansions: The greater melting of #2 and #3 comes at a cost, their thermal expansions are considerably higher, so they will be more likely to craze. Which of these is the best for functional ware? #1, G2926B (left). Its high SiO2 and enough-but-not-too-much B2O3 make it more durable. And it runs less during firing. And does not craze.
A high feldspar glaze is settling, running and crazing. What to do?

This picture has its own page with more detail, click here to see it.
The original cone 6 recipe, WCB, fires to a beautiful brilliant deep blue green (shown in column 2 of this Insight-live screen-shot). But it is crazing and settling badly in the bucket. The crazing is because of high KNaO (potassium and sodium from the high feldspar). The settling is because there is almost no clay in the recipe. Adjustment 1 (column 3 in the picture) eliminates the feldspar and sources Al2O3 from kaolin and KNaO from Frit 3110 (preserving the glaze's chemistry). To make that happen the amounts of other materials had to be juggled. But the fired test revealed that this one, although very similar, is melting more (because the frit releases its oxides more readily than feldspar). Adjustment 2 (column 4) proposes a 10-part silica addition. SiO2 is the glass former, the more a glaze will accept without losing the intended visual character, the better. The result is less running and more durability and resistance to leaching.
Substituting a frit to source B2O3 leads to a dead end

This picture has its own page with more detail, click here to see it.
In the new recipe I am trying to match the oxides (the white, yellow, red, blue boxes). B2O3 is priority, I have supplied enough of the new frit to almost match it, but that far overshoots the Na2O (even if all of the feldspar is removed). While differences in the Al2O3 and SiO2 can be compensated by adjustments in the feldspar, clay and silica, the new frit has far more Na2O and only half the B2O3. The match shown here is the best I am able to achieve, its biggest problem is the significant rise in the calculated thermal expansion, that is going to mean issues with crazing. The only way to use this frit to better match the chemistry of the original glaze would be in combination with another one that has much higher B2O3 and has as little Na2O as possible.
Fix obvious issues in Glazy recipes before even trying them

This picture has its own page with more detail, click here to see it.
Many come to Insight-live after "Glazy recipe fails". A better way is to recognize the potential in a recipe, fix it by recipe logic and calculation (e.g. limit recipes), then try it. Glazy "Red Orange #111576" is an example. It has two things I avoid: Lithium Carbonate (expense) and a high percentage of red iron (slurry gelling). Spodumene is a better source of Li2O, but it contributes lots of Al2O3 and SiO2. We can "make room" for it by replacing the feldspar with Erro frit 3110 (the latter contributes much more sodium and much less Al2O3/SiO2 than the feldspar). Second, use black iron instead of red.
The results using the C6IRED schedule are fabulous. And the cost is way down. Amazingly, black iron does not gel the slurry at all! And it is not nearly as messy as the red. Like any iron red, this has a fluid melt, so is running (although it is applied thickly). The thermal expansion is still quite low, so it should not craze. And the LOI is much lower, which should minimize bubbling.
This titanium blue fails when we switch frits

This picture has its own page with more detail, click here to see it.
The L4655 floating blue recipe is on the outside of the mug. It adds titanium to the GA6-A base. We wanted to reduce the thermal expansion to minimize the likelihood of crazing. So the obvious question was: Could we substitute the Ferro Frit 3134 for Frit 3195 in the base (effectively using GA6-B instead of GA6-A)? The calculation showed that the thermal expansion should drop from 7.6 to 7.2. Unfortunately, it did not work. The two tiles in the front show that (the one on the right adds 2% iron, we thought that might enhance the rutile blue effect). Why did this fail? Likely the raising of the Al2O3 makes the melt stiffer, that is preventing the freedom of movement needed to form the blue crystalline phases.
Use a frit blend ratio to control the amount of kaolin in a glaze recipe

This picture has its own page with more detail, click here to see it.
These are the recipes and calculated oxide chemistries of two pottery glazes (as shown in my account at insight-live.com): The original problem recipe and an adjustment to fix it. Recipe #1 sources boron from a soluble material and three plastic materials are combined to increase drying shrinkage enough to cause cracking when drying (and thus crawling). Recipe #2 solves these problems while producing the same chemistry. It sources boron from two frits (one having almost no Al2O3) whose ratio to each other can be altered to supply more or less Al2O3 to the melt. That enables removing two of the plastics: Ball clay and Gerstley Borate. The remaining 20% EPK is perfect to create a creamy slurry that suspends, applies and dries well.
Why does the inside glaze crystallize with one frit and not the other?

This picture has its own page with more detail, click here to see it.
It is about the oxide chemistry, as shown calculated below the recipes in my account at insight-live.com. These glazes are fired at cone 6 using the C6DHSC schedule (we are focussing on the amber glossy glaze on the insides of the mugs). Most oxides want to form silicate crystals (combine with SiO2) as the glaze cools (if the cooling ramp is slow enough), iron oxide is not the least of these. Alumina (Al2O3) stabilizes the melt, that means it helps the melt to solidify as a glassy solid, not a crystalline one (thus, it does not devitrify). Notice the two Al2O3 values (black-on-red numbers): The glaze on the right has much less. That is because Ferro Frit 3134 contains almost no Al2O3 (notice in the blue panel, only 2%). The alumina in the glaze on the left, sourced more abundantly by Frit 3195, readily releases in the melt, ready to take on its job: Stiffen it and impede the formation of iron silicate crystals during cooling to create a better glass.
Insight-Live comparing a glossy and matte cone 6 base glaze recipe
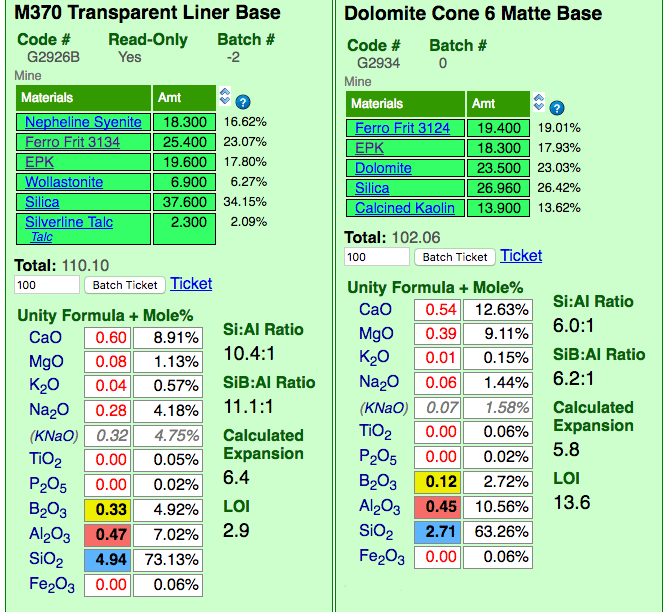
This picture has its own page with more detail, click here to see it.
Insight-live is calculating the unity formula and mole% formula for the two glazes. Notice how different the formula and mole% are for each (the former compares relative numbers of molecules, the latter their weights). The predominant oxides are very different. The calculation is accurate because all materials in the recipe are linked (clickable to view to the right). Notice the Si:Al Ratio: The matte is much lower. Notice the calculated thermal expansion: The matte is much lower because of its high levels of MgO (low expansion) and low levels of KNaO (high expansion). Notice the LOI: The matte is much higher because it contains significant dolomite.
Magnesium carbonate vs. oxide: One big difference

This picture has its own page with more detail, click here to see it.
Here is a screenshot of side-by-side recipes in my account at insight-live.com. It takes 120 mag carb to source the same amount of MgO as 50 mag ox. I just made the two recipes, went into calculation mode and kept bumping up the magcarb by 5 until the chemistry was the same. Note the LOI of the magcarb version is 40. This one would certainly crawl very badly.
Comparing two glazes having different mechanisms for their matteness

This picture has its own page with more detail, click here to see it.
These are two cone 6 matte glazes (shown side by side in an account at Insight-live). G1214Z is high calcium and a high silica:alumina ratio. It crystallizes during cooling to make the matte effect and the degree of matteness is adjustable by trimming the silica content (but notice how much it runs). The G2928C has high MgO and it produces the classic silky matte by micro-wrinkling the surface, its matteness is adjustable by trimming the calcined kaolin. CaO is a standard oxide that is in almost all glazes, 0.4 is not high for it. But you would never normally see more than 0.3 of MgO in a cone 6 glaze (if you do it will likely be unstable). The G2928C also has 5% tin, if that was not there it would be darker than the other one because Ravenscrag Slip has a little iron. This was made by recalculating the Moore's Matte recipe to use as much Ravenscrag Slip as possible yet keep the overall chemistry the same. This glaze actually has texture like a dolomite matte at cone 10R, it is great. And it has wonderful application properties. And it does not craze, on Plainsman M370 (it even survived a 300F-to-ice water IWCT test). This looks like it could be a great liner glaze.
Videos
| Media |
How I Formulated G2934 Cone 6 Silky MgO Matte Glaze Using Insight-Live
I will show you how found a recipe on Facebook, assessed it, substituted my own materials, tested it, adjusted it. |
| Media |
Compare the Chemistry of Recipes Using Insight-Live
Watch me open three recipes side-by-side and turn calculation on for each (in this 19 second video). |
| Media |
A Broken Glaze Meets Insight-Live and a Magic Material
Use Insight-Live.com to do major surgery on a feldspar saturated cone 10R glaze recipe with multiple issues: blistering, pinholing, crazing, settling, dusting and possibly leaching! |
| Media |
Replace Lithium Carbonate With Lithium Frit Using Insight-Live
Raw lithium carbonate can often be replaced with a lithium-containing frit if you can do the chemistry. And you can at insight-live.com. |
| Media |
Predicting Glaze Durability by Chemistry in Insight-Live
How to spot out-of-balance indicators in the chemistry of ceramic glazes that suggest susceptibility to scratching or cutlery marking. |
 |
How I Developed the G2926B Cone 6 Transparent Base Glaze
How I found a pottery glaze recipe on Facebook, substituted a frit for the Gerstley Borate (using glaze chemistry), compared using a melt flow tester, added as much extra SiO2 as it would tolerate, and got a durable and easy-to-use cone 6 clear. |
| Media |
Insight-Live Meets a Silica Deprived Glaze Recipe
This glossy ceramic glaze lacks SiO2 (detrimental to its hardness, leaching resistance, resistance to crazing). Let us see how much silica this glaze will accept! |
| Media |
Getting Frustrated With a 55% Gerstley Borate Glaze
I show you why people love/hate this material and how I substituted it for Ulexite in this crazy recipe to make a far easier-to-use slurry that fires identical. |
| Media |
Analysing a Crazing, Cutlery-marking Glaze Using Insight-Live
A high-nepheline, zero-silica cone 8 silky matte pottery glaze is cutlery marking and crazing. Let's take a closer look and determine why? |
| Media |
Creating a Cone 6 Oil-Spot Overglaze Effect
In this video I use my Insight-live account to do the chemistry to convert an existing MgO-matt glaze into a tin-opacified, high-surface-tension melt for double-layering over a gloss black. I also use it to record my physical testing. |
| Media |
Substitute Ferro Frit 3134 For Another Frit
I use my Insight-live account to do the glaze chemistry to replace Ferro frit 3134 with combinations of three other common Ferro frits. We will see the challenges of doing this in three different types of recipes. |
| Media |
Create a Synthetic Feldspar in Insight-Live
A step-by-step of how to duplicate the chemistry of Minspar by mixing other materials. You will learn the calculate process, the type of testing to do and how to keep track of the results with notes, pictures and links. |
 |
Converting G1214M Cone 6 transparent glaze to G1214Z matte
How I converted a glossy glaze into a matte glaze by using glaze chemistry and recipe logic. |
Links
| URLs |
https://insight-live.com/insight/help
Digitalfire Insight Help WebApp |
| URLs |
https://insight-live.com/insight/help/Recipe+Manager-292.html
Insight-Live Help for the Recipe Manager |
| URLs |
https://insight-live.com/insight/share.php?z=CEavKR6Gye
Insight-live share showing how to fix crazing with cone 6 Leach's Clear |
| URLs |
https://insight-live.com/insight/share.php?z=9TkiB9GPkE
Insight-Live.com share for Worthington Clear with Lead |
| URLs |
https://insight-live.com/insight/share.php?z=uxrGP5yY8P
Insight-Live Project to develop a cone 6 fluid non-crazing clear glaze |
| URLs |
https://insight-live.com/insight/help/It+Starts+With+a+Lump+of+Clay-433.html
Case Study: Testing a Native Clay Using Insight-Live.com |
| URLs |
https://insight-live.com/insight/share.php?z=HzyNzj9ELs
Replacing the Gerstley Borate in recipes containing 50% or more of it Many recipes were built on bases employing exceptionally high percentages of Gerstley Borate. At medium temperatures these melt fluid transparents hosted additions of colorants, variegators and opacifiers. At low fire they were used as transparents over underglaze decoration. |
| URLs |
https://insight-live.com/insight/share.php?z=FuGkW4GNms
Lower Expansion version of G2926B Cone 6 Clear Glaze |
| URLs |
https://insight-live.com/insight/share.php?z=erKwjtZ9tq
G2571A cone 10R silky matte recipe development project |
| URLs |
https://insight-live.com/index.php
Insight-Live.com cloud-based ceramic lab notebook and education platform Ceramic-industry-specific LIMS Lab information management and education system from Digitalfire. This is the software and information to study, understand, adjust and formulate glazes and clay bodies. Replace excel and word documents with a searchable cloud-hosted database accessible from any web browser. View anything side-by-side. Track unlimited specimens and manage large numbers of simultaneous projects. No need to request a quote, just sign up. |
| Articles |
Where do I start in understanding glazes?
Break your addiction to online recipes that don't work or bottled expensive glazes that you could DIY. Learn why glazes fire as they do. Why each material is used. How to create perfect dipping and brushing properties. Even some chemistry. |
| Glossary |
Digitalfire Insight-Live
A cloud-hosted ceramics-targeted LIMS (lab info management system). It does glaze chemistry and physical testing the “Digitalfire way”. For technicians, educators, potters and hobbyists. |
Got a Question?
Buy me a coffee and we can talk

https://digitalfire.com, All Rights Reserved
Privacy Policy

