Contact MeLet's have a Other ways to Support My WorkSubscribe to Insight-Live.com. It is about DIY testing and development, not letting information slip away. Help Me on Social
Login to your online account Chemistry plus physics. Maintain your recipes, test results, firing schedules, pictures, materials, projects, etc. Access your data from any connected device. Import desktop Insight data (and of other products). Group accounts for industry and education. Private accounts for potters. Get started. Download for Mac, PC, Linux Interactive glaze chemistry for the desktop. Free (no longer in development but still maintained, M1 Mac version now available). Download here or in the Files panel within your Insight-live.com account. What people have said about Digitalfire
What people have said about Insight-Live
| June 2026: We are continuing a major code rewrite. Please contact us if you find issues. Thank you. Monthly Tech-Tip from Tony HansenI will send practical posts like these (from thousands I maintain). No ads or tracking. The first email will provide one-click unsubscribe. Signup is being email-bombed by bots. For now, please subscribe inside your insight-live.com account. BlogA transparent glaze is going satin:Is it the feldspar and kaolin substitutions?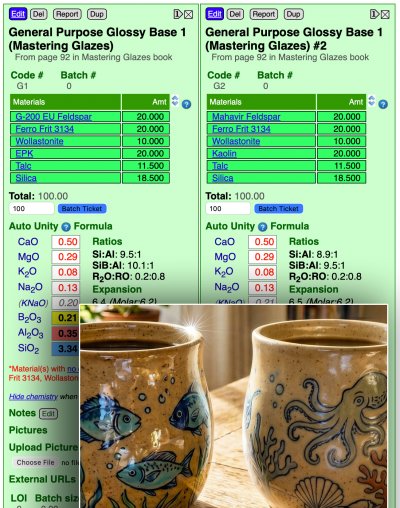
A potter reports that a switch from G-200 feldspar to Mahavir, and EPK to Imerys kaolin, has resulted in this transparent glaze becoming more satin. Is that possible? Yes. Because this glaze is on a unity formula tipping point. Context: MGBase3, Tipping point Thursday 4th June 2026 Alberta Slip as a functional honey-transparent base:The glaze I reach for again and again
The functional surfaces on these pieces all employ the GA6-B base honey glaze recipe. On Plainsman native stoneware clays, especially darker burning ones, typical transparents are very prone to micro-bubble and clouding issues. But not this glaze. The likely reason is that Alberta Slip contains coarser particles (it is only processed to 42 mesh), these act as a fining agent. Context: I drink from these.., This GA6-B glaze is.. Thursday 4th June 2026 Are glazes food safe just because they carry a label?
This Gemini-generated mug could conceivably exist yet carry these labels. Yet experienced ceramic technicians would immediately be suspicious. The glaze is highly fluid and heavily crystallized; both suggest low or very low Al2O3 levels (it is the key oxide that makes glazes durable). If the interior color were produced using a cadmium-containing encapsulated stain, cadmium-release testing would be essential before claiming the ware is food safe. This is clearly engineered for visual effects rather than durability. None of those characteristics prove it is unsafe, but they do mean that labels like "nontoxic" are not substitutes for actual leach testing. A glaze can be made entirely from materials classified as nontoxic and still fail to meet the durability standards expected of functional foodware. Context: Commercial glazes on decorative.., ASTM D-4236 - Standard.. Thursday 4th June 2026 Should I glaze the outside of this mug now? No!
This bisque mug has been glazed on the inside. But the bisque has absorbed water from that glaze, and this thin-walled mug is now waterlogged as a result (except at the thicker base). It does not have the absorbency needed to build up a thick enough layer of glaze on the outside. Even if it did, the water from the two glazes would wet the bisque so much that its drying time would be greatly extended. This is a problem because the mechanism of attachment of glaze to the body is fragile and works best when the glaze dries quickly. When drying is too very slow, bubbling and cracking often occur (leading to crawling in the firing). Context: Does bisque ware need.., Glaze thickness Monday 1st June 2026 Yixing teapot making. Is it magic?Or highly evolved craft and science?
The Yixing teapot craftsmen appear to break all the rules and yet produce impossibly delicate and symmetrical pieces. Hao-Tong Yan, one of those craftsmen, and I have been trying to understand the technical reasons for how this amazing craft is possible. It turns out not to be magic, but actually a highly evolved understanding of a very unusual material. Here are some of the things that we are coming to understand (which is making it possible to create a facsimile of the clay in North America). Context: Yixing Clay Teapots on.., Stunning video of Yixing.. Saturday 30th May 2026 2026: ChatGPT is doing credible troubleshooting
I uploaded a screenshot of this recipe panel from Insight-live and asked ChatGPT why sanitaryware using this glaze is dunting. Its response is impressive, good enough to provide remediation ideas. Context: ChatGPT was completely wrong.., ChatGPT is surprisingly wrong.., Two ChatBots square off.., Does ChatGPT know the.., Sanitary ware Tuesday 26th May 2026 Crystalline glazes normally craze:Here is one way to fix that
The mug on the left, made by Holly McKeen, is a typical cone 10 Grolleg kaolin mullite porcelain (highly vitrified, low in residual quartz). Its glaze is crazed. Crystalline glazes are high in Na2O, making crazing virtually certain. Since most pieces are decorative, crystal glazers just accept this as part of the process. But these are functional mugs, the glaze needs to fit (if only for ware strength). Context: Crystalline glazes, Calculated Thermal Expansion Tuesday 26th May 2026 Incorrect craze-fixing advice is still common online:Well demonstrated using an AI-generated photo!
Crazing is one of the most common glaze defects. AI image generators can produce this really convincing photo, but AI explanations often still recycle oversimplified glaze-fit advice from the web. Let's work in reverse to see why using this speckled stoneware, it has lots of ball clay and quartz, it is easy to fit glazes to. What would it take to craze the glaze on the right? A lot. Glazes that craze out of the kiln on quartz-rich bodies are not "slightly misfit"; they are "hugely misfit". Under or over-firing, or holding less time at temperature, would not be enough to craze it. Reducing the silica enough to start severe crazing like this would fundamentally alter the glaze character and functionality. Context: Glaze Crazing Sunday 24th May 2026 Here is what dipping engobes can do:Go on even. In one coat. Stay put.
When you learn to make and use engobes correctly, they make magic possible. Here I am turning a dark rustic body into a smooth white one (rear mugs) and a white body into a dark one (front). The engobes have been applied at the leather-hard stage. That is the perfect time, the engobe and body are clay bodies, designed to fit each other; they dry together and fire together creating an inseparable bond. Context: How stop dripping and.., Here s how we.., Why your supplier does.., Why your supplier does.. Thursday 21st May 2026 Add 6% lithium carbonate to an Alberta Slip glaze:And this happens!
Left is G3933A, it is an 80:20 mix of our matte and glossy cone 6 base recipes (plus a mix of iron oxide, tin oxide and rutile). The body is Plainsman Coffee Clay. Because of repeated issues with crawling a project was started to create the same effect using Alberta Slip to supply as much of the chemistry as possible. Along that road, the opportunity arose to add lithium (to duplicate Amaco PC-32, a classic Albany/Lithium recipe). That is the glaze on the mug on the right, G3933G1, it has 6% lithium carbonate. Lithium is a super powerful melter, turning this into a very reactive glaze! To make a 500ml jar of brushing glaze, in 2023, required about $7 worth of lithium carbonate. Context: Lithium Carbonate, GA6-G, Oatmeal glazed mugs, Amaco achieves the stunning.., Recognize these universal oxidation.. Wednesday 13th May 2026 |

https://digitalfire.com, All Rights Reserved
Privacy Policy








