Material Substitution
Material substitutions in ceramic glaze and body recipes must consider their chemistry, mineralogy and physical properties
Key phrases linking here: material substitution - Learn more
Details
Material substitution is a constant and ongoing part of any ceramic enterprise that is using clay and/or glaze recipes. Often lack of availability, quality issues and price are the motivating factors. In addition, when recipes need to be used in another locality where the same raw material brands or even types are unavailable, substitutions must be done.
In ceramics, glazes fire the way they do primarily because of their chemistry. The most basic substitution is to simply replace one material for another which has a chemistry similar enough that the fired properties will not be adversely affected. This is often possible with different brand names of the same mineral or refined material (e.g. Calcium carbonate, zinc oxide). The more complex the chemistry of the raw material, the more likely there is to be issues with changes to another (e.g. feldspar). In some cases substitution recipes are recommended where a mix of two or more other materials is said to be equivalent. Another problem can occur where, although the chemistry of the substitute is very similar, it's physical properties or particle size are different enough to affect the working properties of the glaze (e.g. a kaolin) or even it's fired appearance (a metal oxide).
A given chemistry can be supplied by many different mixes of refined and raw material powders, each of these having advantages and disadvantages regarding price, ease of use in production, toxicity, etc. It is common to use glaze chemistry to calculate how to juggle a recipe to substitute one material for another of slightly or even very different chemistry while maintaining the chemistry of the glaze as a whole. The calculation is more complex where the substitute is bringing along other oxides not in the material being substituted (or fewer) or the original or substitute has a very different weight loss on firing (LOI). Frits especially can have complex chemistries and obviously it is more complicated if they are involved in the substitution calculation.
When clay bodies and porcelains require material substitution, the issue is physical properties (which are often not directly related to chemistry). Thus testing must be done to see how maturity, drying properties, plastic and/or forming behavior, texture, firing color and character, thermal expansion and other properties are affected. A series of tests usually must be done to alter the recipe to accommodate the new material while maintaining the needed properties.
Related Information
Comparing two brands of calcium carbonate

This picture has its own page with more detail, click here to see it.
This GLFL test compares the melt flow, at cone 6, of two glaze recipes containing the calcium carbonates (of which they make up 27%). Notice the amount of bubbles (due to the high LOI, or loss on ignition). The 3HX is flowing a little more. This could be because of a difference their proportions of dolomite and calcium carbonate minerals, the individual mineral purities or the particle size (or all three). Whatever the case, 3HX will make a glaze flow a slightly better.
Testing two brands of tin oxide in a melt flow tester

This picture has its own page with more detail, click here to see it.
This melt fluidity tester compares two different tin oxides in a cone 6 transparent glaze (G2926B). Opacifiers affect not just opacity in glazes, but also liquid properties of the melt. The length, surface character, opacity and color of these flows provide an excellent indication of how similar the two materials are. This is the GLFL test.
Testing a new brand of dolomite

This picture has its own page with more detail, click here to see it.
Dolomite is a key material for glazes, especially mattes. We were forced to adopt a new brand and needed confidence it was equivalent. Three tests were done to compare the old long-time-use material (IMASCO Sirdar) with a new one (LHoist Dolowhite). The first melt flow tester compares them in a very high dolomite cone 6 recipe formulated for this purpose; the new material runs just slightly more. The second tester is uses the G2934 cone 6 magnesia matte recipe with 5% black stain; the new material runs a little less here. The third test is the high dolomite glaze on a dark burning clay to see the translucency and compare the surface character. They are very close. These three gave us the confidence to proceed.
L3617 Cornwall Stone substitute vs. real Cornwall Stone

This picture has its own page with more detail, click here to see it.
This melt fluidity comparison demonstrates how similar the substitute L3617 recipe (left) is to the real material (right). 20% Frit 3134 has been added to each to enable better melting at cone 5 (they do not flow even at cone 11 without the frit). This substitute is chemically equivalent to what we feel is the best average for the chemistry of Cornwall Stone.
Laguna Barnard Slip substitute fired bars

This picture has its own page with more detail, click here to see it.
These are fired bars of Laguna SG758 Barnard Slip substitute going from cone 04 (bottom) to cone 6 (top). It is melting at cone 6. The bars are expanded above cone 6 and becoming quite porous. The drying shrinkage is around 7%, it is quite plastic.
G2934 using Fusion Frit F-19 instead of Ferro 3124

This picture has its own page with more detail, click here to see it.
G2934 is a popular recipe and there has been alarm recently because of the difficulty in getting the Ferro frit and the variation in its quality in recent years. This motivated us to get a supply of the Fusion equivalent, F-19. When doing substitutions like this we do testing in glazes and with melt fluidity tests - like this GLFL test.
Grolleg vs New Zealand - Which kaolin is better for translucency?

This picture has its own page with more detail, click here to see it.
The NZK body, Polar Ice, is on the left. The Grolleg one, L3778D, is on the right. They are not the same recipe, the feldspar content in the L3778D has been adjusted to match the degree of vitrification (Grolleg contains some feldspar). Clearly, the NZK has better translucency. And it fires whiter.
The difference between Silica 90 and Silica 45 will affect the glaze melt

This picture has its own page with more detail, click here to see it.
Quartz particles have a high melting point, they must enter the glaze melt by being dissolved by it (usually the last particles to do so). Obviously, the silica should be as fine as possible to increase its surface area to be more readily dissolved. The more that dissolves the closer the physical properties of the fired glaze will be to the theoretical (e.g. degree of melting, thermal expansion, transparency, durability). This brand of silica, #90 classifies as 200 mesh even though 2.8% remains on the 200 mesh screen. Not surprisingly, their #45 grade retains 1.9% on the 325 mesh screen. However, the most significant aspect is how much of the #90 is on the 325 and 270 mesh screens: 26%. The #45 grade only retains 2.6% on them! This is a huge difference and shows the value of using the finer material. It would take a typical ball mill hours to make this difference.
Substitute Ferro Frit 3134, using glaze chemistry, in three glaze types

This picture has its own page with more detail, click here to see it.
Can't get frit 3134 for glaze recipes? Can you replace it with frit 3124? No, 3124 has five times the amount of Al2O3 (the second most important oxide in glazes) and half the amount of B2O3 (the main melter). This ten-minute video presents a glaze chemistry approach that is easier to do than you probably think. It deals with three different glaze recipe types lacking sufficient clay to suspend the slurry. Learn to source the needed oxides from two other Ferro frits, 3110 (or Fusion F-75) and 3195 (Fusion F-2), and end up with at least 15% kaolin in each. A unique approach is required in each situation. Two of the calculations produce improved slurry properties and one yields a recipe of significantly lower cost. If you have a recipe that needs this and need help please contact us.
Is Ferro Frit 3124 a viable substitute for Frit 3134?

This picture has its own page with more detail, click here to see it.
This is a GLFL test comparing the melt flow of the three materials at 1800F. Frit 3124 is barely out of the starting gate and the other two have crossed the finish line! With frits chemistry is a big deal, they are all about supplying oxides to the melt. Frit 3134 is low-alumina/high-boron, 3124 is medium-alumina/low-boron and 3195 is medium-alumina/high-boron. Boron is the melter. Alumina thickens the melt and hardens the glass. Just from this it appears that Frit 3195 is a better starting point for calculations to replace frit 3134.
Calculating a substitute for Minspar

This picture has its own page with more detail, click here to see it.
Why do this? We did not have it in stock and customers needed to mix recipes. When the chemistries of the two feldspars are very similar substitution is often not a problem, especially when a recipe only calls for 5 or 10%. However, when a recipe calls for a significant percentage the situation becomes much trickier (in our cone 6 test recipe, "Perfect Clear", 40% Minspar is needed). Feldspars are almost a glaze in themselves, just needing silica and alumina to shift their chemistry toward 'glazedom'. In this project I calculated a mix of materials, in my Insight-live.com account, that sources the same chemistry as Minspar. I made cone 6 GLFL tests comparing the pure Minspar and Minspar substitute (left) and comparing the Perfect Clear glaze with each feldspar (right). As you can see, the similarity in melt flow is stunning! This is a good demonstration of just how practical and valuable glaze chemistry calculation can be.
Which is the champion melter of American/Canadian feldspars?
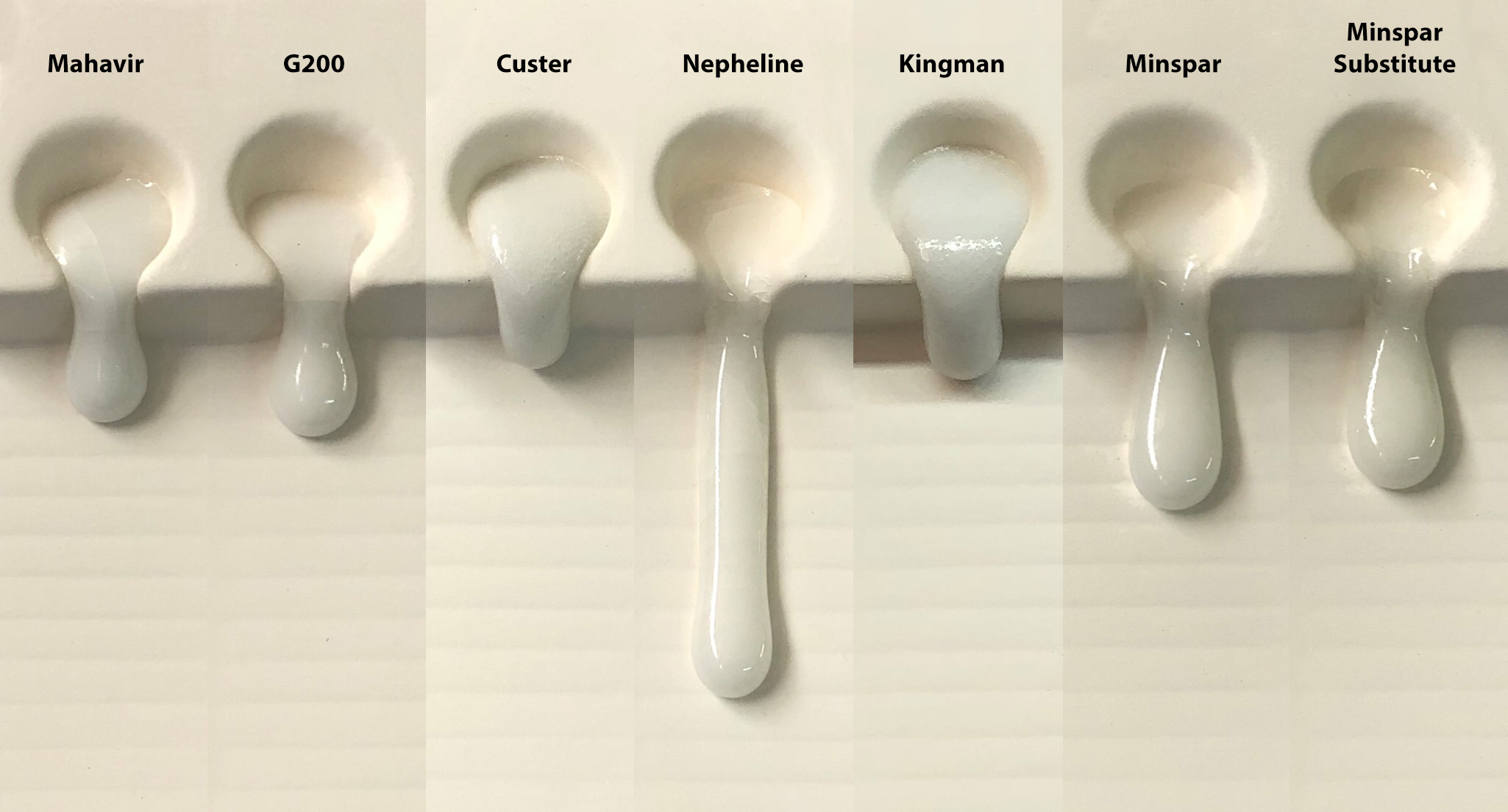
This picture has its own page with more detail, click here to see it.
Feldspars are employed in glaze recipes as melters. So comparing their melt fluidities should be helpful in deciding if one can substitute for another (of course, if possible, a soda-predominant feldspar should be substituted for another soda spar). Feldspars don't melt alone at cone 6 (2200F) so we mixed each with 15% Ferro Frit 3195 (we consider a feldspar a material that sources KNaO). Nepheline Syenite, this is A270, is the champion melter here. Other similar ones can be spotted easily. In the end, degree of melt is a valid consideration in determining if one feldspar is a viable substitute for another in a recipe. Even if the feldspar you want to substitute does not melt as much a little frit can be added to the recipe to make up for the difference (e.g. 3-5%).
Arbuckle Majolica glaze using Fusion Frit F-19

This picture has its own page with more detail, click here to see it.
Fusion Frit F-19 is said to be a substitute for Ferro Frit 3124, for this particular glaze that appears to be true. The Arbuckle Majolica glaze was applied on Plainsman L210 and fired at cone 04 (but not as thickly applied as would be customary on Majolica ware). On the left is the one with Ferro Frit 3124, on the right is the one with the F-19. The recipe is based on Ferro Frit 3124 and adds 20% of a feldspar neheline mix (likely to increase thermal expansion because it shivered on the original clay body). It is suspended by 10% kaolin. This may craze on your clay body, the G1916Q thermal expansion adjustable transparent base might be a better solution (it responds similarly to a zircon/tin addition to opacify).
Links
| Glossary |
Ceramic Material
Ceramic materials are employed in the ceramic industry to make glazes, bodies, engobes and refractories. We study them at the mineral, chemical and physical levels. |
 PayPal | No tracking, No ads, No paywall, No transient content! Just organized, concise information constantly updated and improved. Was this helpful? Consider supporting me. |
| By Tony Hansen Follow me on        |  |
Got a Question?
Buy me a coffee and we can talk

https://digitalfire.com, All Rights Reserved
Privacy Policy
