Phase Separation
Phase separation in glaze melts creates microscopic discontinuities that affect transparency, color variegation, matte surfaces, and reactive glaze effects.
Key phrases linking here: phase separations, phase separation, phase changes - Learn more
Details
Phase separation occurs when a molten glaze does not remain a single chemically uniform liquid but separates at the microscopic scale into two or more glassy phases of slightly different composition. During cooling, these phases may form droplets, interconnected networks, or domains dispersed within one another. Although the glaze appears solid after firing, the final glass may contain countless microscopic discontinuities created during the melt or cooling process.
These discontinuities arise because certain oxide combinations are only partly compatible in a silicate melt. Instead of remaining fully dissolved in one homogeneous glass, portions of the melt separate into regions richer in particular oxides. This often develops late in firing or during cooling, when melt mobility decreases and local chemistry differences become more significant. In some cases, incomplete dissolution of batch materials or delayed interaction between decomposing particles can encourage the process.
Colorants and opacifying oxides may preferentially concentrate in one phase rather than another. As a result, phase separation can produce variegation, clouding, soft mottling, or subtle color shifts because different phases may have different refractive indices, viscosities, and capacities to host coloring ions.
Phase separation is often undesirable in glazes intended to be crystal clear. The internal interfaces between phases scatter light, reducing transparency and producing milkiness or haze even when no crystals are present. This is one reason some high-boron transparent glazes appear slightly cloudy despite being fully melted.
In industrial glassmaking, phase separation is carefully controlled because it affects optical clarity and durability. Increased alumina often helps stabilize the glass network and suppress separation, although excessive alumina can also promote devitrification if cooling conditions allow crystal growth. Sodium can improve melt homogeneity by increasing fluxing action, but excessive Na₂O also raises thermal expansion and may create fit problems in ceramic glazes.
In ceramics, however, phase separation is not always undesirable. Many so-called reactive glazes depend on controlled microscopic heterogeneity to develop visual depth and complexity. Sanitary ware glazes, such as those used on sinks and toilets, are formulated to remain highly homogeneous, producing smooth, durable, fully transparent glass with minimal internal discontinuity.
Some matte glazes are partly influenced by phase separation. In dolomite mattes, for example, MgO encourages limited immiscibility in the melt and can also promote crystal development during cooling. The matte effect usually results from both microscopic phase separation and fine crystal precipitation rather than phase separation alone.
Titanium and rutile additions strongly encourage discontinuity because they reduce glass uniformity and often stimulate crystal nucleation. In fluid iron-bearing glazes, rutile can intensify local segregation of iron and titanium, contributing to streaking effects such as blue rivulets suspended in amber glass. In these cases, the visual effect often involves both phase separation and selective crystallization acting together.
Related Information
Phase separation close-up
The power of modern phone cameras
This picture has its own page with more detail, click here to see it.
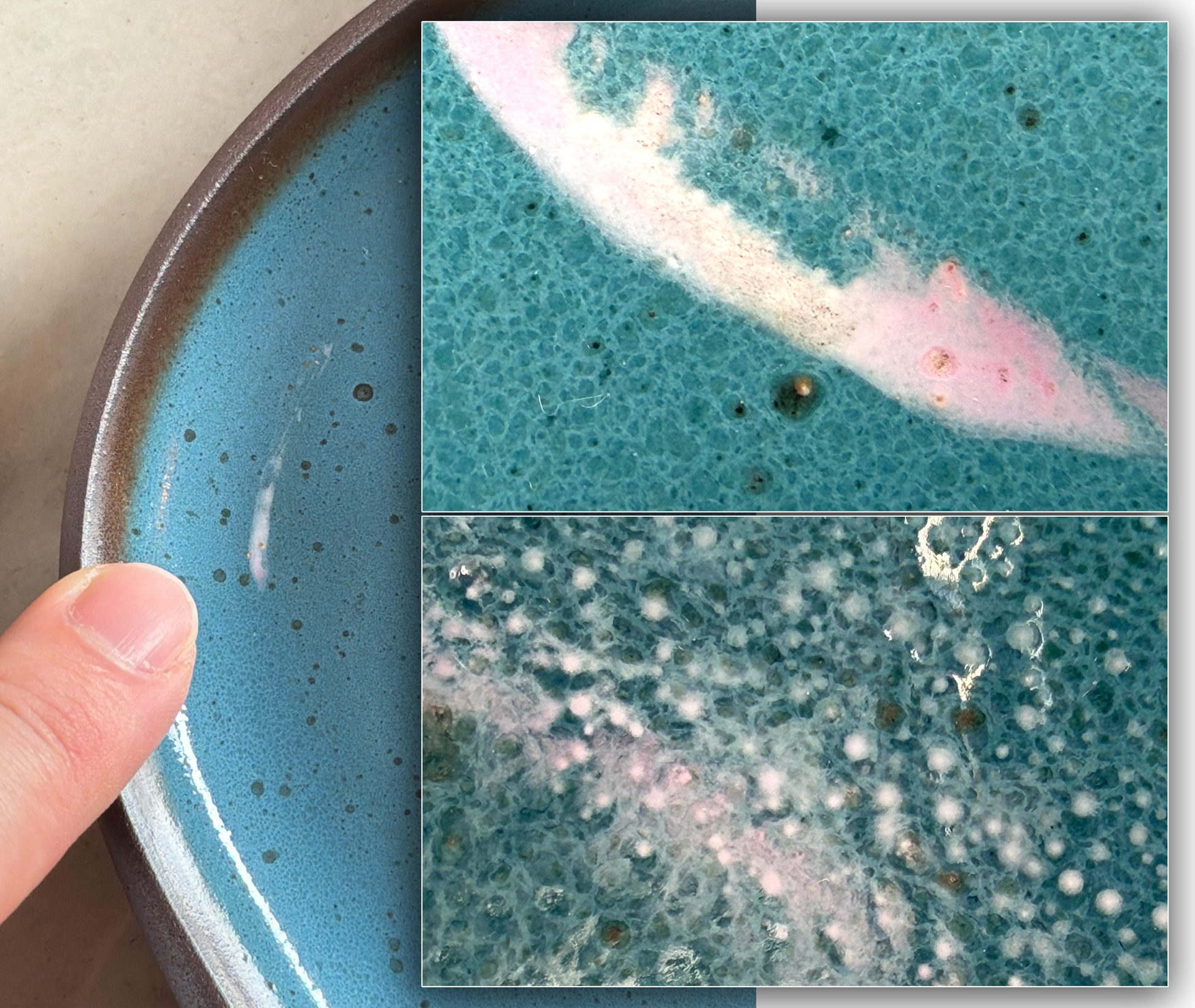
This reduction stoneware glaze is producing white streaks on some pieces (left center). The body is a coarse iron stoneware. A magnification is needed to better explain this.
It is 2025, many smartphones now have dedicated macro lenses and can be held as close as a 1 centimeter. They automatically sense placement and switch to using the macro lens. Of course, the phone must be held rock steady and good lighting is essential. If you are a doubter of what they can produce, look at the two magnifications on the right. On the top one, the white streak is clearly visible, floating in a sea of phase-separated glass patterned by earlier-escaping bubbles. The extreme magnification on the bottom right appears to implicate tiny crystals growing in an area where late bubbles have escaped, changing the pattern of phase separation. This doesn’t yet explain the cause, but it is valuable information courtesy of a macro lens.
A glaze whose visual effect is partly a product of phase separation

This picture has its own page with more detail, click here to see it.
Example of a rutile-iron stained glaze. Rather than crystallizing to form the visual effect, the rutile is forming a phase separation that produces the streaking blue in the amber background glass.
Variegation and phase separation with about 5% rutile

This picture has its own page with more detail, click here to see it.
The glaze is a dolomite matte fired to cone 10R. High fire reduction is among the best processes to exploit the variegating magic of rutile.
Micrograph of phase separation in a glaze

This picture has its own page with more detail, click here to see it.
Two glazes of the same chemistry:
But supplied by different sets of materials

This picture has its own page with more detail, click here to see it.
These two specimens are the same terracotta clay fired at cone 03 in the same kiln. The glaze on the left combines 30% frit with five other materials. The one on the right mixes 90% frit with one other material (kaolin). Ulexite is the main source of boron in #1, it decomposes during firing, expelling 30% of its weight as gases, mostly CO2, forming micro-bubbles. Each of the six materials in that recipe has its own melting characteristics; their interaction creates a non-homogeneous glass containing phase separations (discontinuities) that affect the fired surface. In the fritted glaze, by contrast, all the particles soften and melt in unison and produce no gas. Notice that it has also interacted with the body, fluxing and darkening it (thus forming a better interface). And it has passed (and healed) more of the bubbles from the body.
Links
| Glossary |
Crystalline glazes
A type of ceramic glaze made by potters. Giant multicolored crystals grown on a super gloss low alumina glaze by controlling multiple holds and soaks during cooling |
| Glossary |
Matte Glaze
Random material mixes that melt well overwhelmingly want to be glossy, creating a matte glaze that is also functional is not an easy task. |
| Glossary |
Reactive Glazes
In ceramics, reactive glazes have variegated surfaces that are a product of more melt fluidity and the presence of opacifiers, crystallizers and phase changers. |
| Glossary |
Magnesia Matte
Magnesia matte ceramic glazes are “microstructure mattes” while calcia mattes are “crystal mattes”. They have a micro-wrinkle surface that forms from a high viscosity melt and microscopic phase separation, both of which prevent levelling on freezi |
| Glossary |
Melting Temperature
The melting temperature of ceramic glazes is a product of many complex factors. The manner of melting can be a slow softening or a sudden liquifying. |
| Glossary |
Rutile Blue Glazes
A type of ceramic glaze in which the surface variegates and crystallizes on cooling in the presence of titanium and iron (usually sourced by rutile) |
| Recipes |
G2571A - Cone 10 Silky Dolomite Matte glaze
A cone 10R dolomite matte having a pleasant silky surface, it does not cutlery mark, stain or craze on common bodies |
| Properties | Glaze Variegation |
| Articles |
High Gloss Glazes
A transcript of a presentation at the 3rd Whitewares conference at Alfred University in the spring of 2000 by Richard Eppler. |
 PayPal | No tracking, No ads, No paywall, No transient content! Just organized, concise information constantly updated and improved. Was this helpful? Consider supporting me. |
| By Tony Hansen Follow me on        |  |
Got a Question?
Buy me a coffee and we can talk

https://digitalfire.com, All Rights Reserved
Privacy Policy
