Transparent Glazes
Every glossy ceramic glaze is actually a base transparent with added opacifiers and colorants. So understand how to make a good transparent, then build other glazes on it.
Key phrases linking here: transparent glazes - Learn more
Details
A fully transparent glaze is simply one that does not have opacity. But there are degrees of transparency. For example, if a glaze is matte it may show the color of the underlying body and decoration, but these will be muted (so it is actually translucent). Completely transparent glazes look like a glass container or a mirror, perfectly clear and glassy smooth. Glazes that you might have always taken to be transparent may appear much less so when compared side-by-side with a true brilliant glossy clear. It is quite difficult to achieve complete transparency. Various factors can cause light to scatter (reflect) from discontinuities or surfaces on or within the glass matrix. These include entrained bubbles (or surface disruptions caused when they break the surface), bodies containing impurities, phase separation, crystal growth during cooling (devitrification), unmelted or undissolved particles (e.g. silica), agglomerations or simple lack of a smooth glassy surface. Changes in the chemistry, materials, application method and firing may all be needed to deal with these factors. Low LOI materials produce the least gases during firing. Frits melt better and phase-change less. Low water content glazes have the most dense, bubble-free laydown. Hold times on firing (either a top-soak or drop-and-soak) help smooth out the surface. Certain chemistries are susceptible to crystallization, especially if cooling is too slow. Fine-particled bodies made from clean materials, glazes that are ball-milled well and applied as thinly as practical and densed laydown as possible will also give best results.
The most common use of transparent glazes is over underglaze decoration. Anyone using this process will tell you how delicate it is. Underglaze colors commonly bleed at the edges, repel or crawl the overglaze or generate micro-bubbles that cloud the otherwise transparent glass. The process of applying the glaze, getting it just the right thickness and firing it to the right degree is as important, or more important, than the recipe of the transparent glaze. The difficulty of getting it working could test your will! Your supplier may have combinations of a clay body, color, an overglaze and procedures that are likely to work. Generally, lower temperatures offer easier solutions.
The most brilliant transparents are often high in Na2O and K2O (unfortunately these oxides contribute to a high thermal expansion and crazing). Glossy transparents also usually have a high SiO2:Al2O3 ratio (greater than 10).
When transparent glazes are coloured, as opposed to opaque colors, variations in glaze thickness produce color highlighting (celadon glazes are a classic example). But many glazes can also change color with thickness, this happens because of the tendency to crystallize on cooling coupled with the fact that crystal growth often varies with glaze thickness (fluid melt high-iron glazes do this if cooled slowly enough).
Transparents can amplify the coloring effect of iron in an underlying body (they flux the body surface and make it more mature). For example, at cone 6, a porcelain or white stoneware may appear more yellowish under a transparent than without a glaze covering. One method to deal with this is add 0.05 to 0.1% blue stain (to the body). Transparents also affect the color of underlying brown and red bodies. While the transparent glazed surface of low-fire terra cottas may burn to a pleasant red color at cone 06-04, at cone 02 the glaze vitrifies the surface to fire brown (middle-temperature red bodies suffer the same fate).
When a transparent is intended as a base one must consider the types of visual effects desired. Typical functional transparents may melt to a smooth glossy glass as-is, but when certain colorants or opacifiers are added the brilliance of the surface may be lost somewhat. For this reason, the melt fluidity must be taken into account (more fluid melts will stay fluid while hosting refractory colorants and opacifiers). However, they also present more of a danger of running, crazing and leaching.
Related Information
G2926B cone 6 transparent liner glaze: Proven, reliable, durable

This picture has its own page with more detail, click here to see it.
While colorful and layered glazes on the outsides of pieces get lots of praise and glory, transparent or white glazes providing the functional surface on the insides of pieces often get little attention from potters. Really, what good is an attractive piece if the food surface is crazing, blistering, leaching or cutlery marking? Or if it converts the piece into a time bomb? This cone 6 liner glaze, G2926B, is an example of how I found a glaze, recognized its potential and then adjusted the recipe to resist crazing on our clay bodies, fire durable and leach resistant and act as a base to host colorants, opacifiers and variegators. I get the best fired results using the C6DHSC firing schedule and very good performance as a dipping glaze when the slurry is thixotropic. One of the reasons this recipe is so widely used is that it is well-documented, having a code number that Google indexes. Drinking from a mug having a quality and fitted functional surface and a nice crisp line dividing the outside and inside glazes instills pride in me as the maker. Watch the G2926B video to see how I developed this. What is the outside glaze? It is the G2934Y matte base recipe plus 8% Mason 6027 stain. The clay is MNP which I make myself.
Transparent glazes often work poorly on dark stoneware bodies

This picture has its own page with more detail, click here to see it.
These are fired in cone 6 oxidation. They are all the same clay body (Plainsman M390). The center mug is clear-glazed with G2926B (and is full of bubble clouds). This dark body is exposed inside and out (the other two mugs have L3954B white engobe inside and midway down the outside). G2926B clear glaze is an early-melter (starting around cone 02) so it is susceptible to dark-burning bodies that generate more gases of decomposition - they produce the micro-bubble clouding. That being said, the other two glazes here are also early melters - yet they did not bubble. Left: G2926B plus 4% iron oxide. That turns it into an amber color but the iron particles act as a fining agent (vacuuming up the bubbles)! Right: Alberta Slip GA6-B. It also fires as an amber-coloured glass, but on a dark body, this is an asset.
Why dipping transparent glazes may not cover underglazes well

This picture has its own page with more detail, click here to see it.
Underglaze brushstrokes were applied to this test cup at the leather hard stage (lower left) and then bisque fired on. On the lower right a ball of the pure underglaze emerged from the same bisque firing, notice that it is certainly fusing enough to seal the surface of the bisque. Notice what happened on the upper left: Although the bisque piece was immersed in a dipping glaze the underglaze is not covered. Yet on the upper right a just-applied transparent brushing glaze has covered well (although three coats were needed with plenty of drying time between). Why is this? Brushing underglazes contain a lot of CMC gum, it makes them spread well like paint - and dry hard (but also denser and less absorbent). Manufacturers of brushing underglazes assume that gummed brushing glazes will also be used over their products. How can coverage be achieved using a DIY transparent? Mixing your glaze as a thixotropic slurry, a first coat dipping glaze or even as a brushing glaze.
The right transparent glaze makes this underglaze design shine!

This picture has its own page with more detail, click here to see it.
The underglaze decoration on this cone 6 porcelain plate, by Dana Powell @danaspottery, is undeniably impressive. But the transparent overglaze is equally amazing. Why? It is not easy to get adequate transparency, crisp brushstroke edges and a defect-free surface at cone 6. While a number of factors are at play, perhaps the most important is a controlled thickness and proper adherence and coverage over the underglaze brush strokes (true for any recipe). Dipping glazes tend to go on too thick on the porcelain and don't stick well to underglazes (underglazes are gummed and, when dried, have almost no porosity to adhere an overglaze). A commercial low-specific-gravity gummed brushing glaze is an option; layers go on thinly, enabling careful control of thickness (of course, it can be time-consuming to paint all the layers needed). However, the brushing action can smear the underglaze. Mixing the transparent as a base-coat dipping glaze could be the best answer. With the right amount of gum and water (by experimentation) it will drain fast enough to dry in a minute or so and spread out evenly into a thin, dense and even laydown of the right thickness, one that adheres equally to body and underglaze (of course, a method is needed to hang the draining plate on edge).
Underglazes require a fluid melt transparent
But extra melt fluidity comes at a cost

This picture has its own page with more detail, click here to see it.
These porcelain mugs were decorated with the same Amaco Velvet underglazes (applied at leather hard), then bisque fired, dipped in clear glaze and fired to cone 6 (using a drop-and-hold schedule). While the G2926B clear glaze (left, A) is a good glossy transparent for general use, its melt fluidity is not enough to clear the LOI micro-bubble clouds that dull the colors below (strangely, underglaze colors can generate them too). However, the G3806C recipe (right, B) has a more fluid melt, which is one factor that better enables bubble escape. Its melt also has a lower surface tension. An additional factor is thickness. A thinner layer of B would be even better (brushing glazes enable thin layering).
But B has downsides. Running is one, but not a factor here because we want it thin. Its high flux content also means it fires less durable. And, its high KNaO content raises the thermal expansion (COE) and thus the danger of crazing. Although it fits this porcelain, it crazes on others.
Underglaze decoration at low, medium and high temperature reduction

This picture has its own page with more detail, click here to see it.
Left is Plainsman Zero3 stoneware fired at cone 03. Middle is Polar Ice fired at cone 6d. Right is Plainsman P600 fired at cone 10R. The same black and blue underglazes are used on all three, but each has its own transparent glaze (left G2931K, middle G3806C, right G1947U).
Transparent glaze quality on terra cotta is about multiple things

This picture has its own page with more detail, click here to see it.
Glaze chemistry, slurry preparation and application thickness. These two mugs are made from the same clay, Plainsman Snow. Amaco V-303 Terracotta underglaze has been applied to each. These two glazes have the same chemistry. But the one on the left was mixed as a dipping glaze, from a recipe, then slurried and then sieved at 80 mesh. However, the sieve did not break up the agglomerates of Veegum and New Zealand kaolin. This problem was resolved by mixing batches of the glaze in a kitchen blender at top speed. The glaze on the right is Spectrum 700, a commercial brushing glaze - it has been vigorously mixed and sieved as part of their production process. There is a second issue here also: Thickness. The mug on the left was dipped and held in too long, getting it on too thick. On the right, the gummed glaze was painted on - three thin coats were evenly applied, enabling careful control of thickness.
Vitreous red body color with a clear glaze at cone 6. Impossible.

This picture has its own page with more detail, click here to see it.
The two back pieces are traditional Mexican terra cotta ware. Lead glazed, porous, not durable at all. The front two pieces are stoneware, produced by two Mexican manufacturers to emulate the look of traditional ware. Both tried. Both failed. It is pretty well impossible to clear-glaze a mid temperature red burning stoneware and get a red color. The glaze fluxes the clay surface and turns the color to the more vitrified version of the body, which is always brown. The terra cotta is fired so far below the temperature at which the clay vitrifies to brown that it maintains the red body color. To get the color in the stoneware plate on the left they brushed on a red-stained engobe (you can see the brush strokes). This worked. And the glaze has fired crystal-clear (likely because they bisque fired it very high). But there is a problem. Although not crazed when purchased, it crazed badly after several months. The bowl on the right also has a beautiful transparent glaze and it has maintained crisp edges on the decoration (and with no micro-bubble clouding). But this is as dark as they could make the clay without having it turn brown under that clear glaze.
Two base clear glazes with 2% copper:
One is bubbling and one is not.

This picture has its own page with more detail, click here to see it.
By itself, without copper, the G2926B recipe (right) produces a better and more durable glass (comparing the cups in the back). But a 2% copper addition, front, turns its surface to a mass of unhealed bubble-escapes. The G3808A recipe, on the left, develops much more melt fluidity, the extra mobility enables the bubbles, created by the decomposing copper, to coalesce, grow, break at the surface and heal before the melt stiffens too much.
In pursuit of a reactive cone 6 base that I can live with

This picture has its own page with more detail, click here to see it.
These GLFL tests and GBMF tests for melt-flow compare 6 unconventionally fluxed glazes with a traditional cone 6 moderately boron fluxed (+soda/calcia/magnesia) base (far left Plainsman G2926B). The objective is to achieve higher melt fluidity for a more brilliant surface and for more reactive response with colorant and variegator additions (with awareness of downsides of this). Classified by most active fluxes they are:
G3814 - Moderate zinc, no boron
G2938 - High-soda+lithia+strontium
G3808 - High boron+soda (Gerstley Borate based)
G3808A - 3808 chemistry sourced from frits
G3813 - Boron+zinc+lithia
G3806B - Soda+zinc+strontium+boron (mixed oxide effect)
This series of tests was done to choose a recipe, that while more fluid, will have a minimum of the problems associated with such (e.g. crazing, blistering, low run volatility, susceptibility to leaching). As a final step the recipe will be adjusted as needed. We eventually evolved the G3806B, after many iterations settled on G3806E or G3806F as best for now.
An ultra-clear brilliantly-glossy cone 6 clear base glaze? Yes!

This picture has its own page with more detail, click here to see it.
I am comparing 6 well known cone 6 fluid melt base glazes and have found some surprising things. The top row are 10 gram GBMF test balls of each melted down onto a tile to demonstrate melt fluidity and bubble populations. Second, third, fourth rows show them on porcelain, buff, brown stonewares. The first column is a typical cone 6 boron-fluxed clear. The others add strontium, lithium and zinc or super-size the boron. They have more glassy smooth surfaces, less bubbles and would should give brilliant colors and reactive visual effects. The cost? They settle, crack, dust, gel, run during firing, craze or risk leaching. Out of this work came the G3806E and G3806F.
One way for an ultra clear at low fire: Magnesia-alkali, low Si:Al ratio, more boron.

This picture has its own page with more detail, click here to see it.
On the left is G2931J, a zinc alkali fluxed and high Si:Al ratio glaze. Those look like micro-bubbles but they are much more likely to be micro-crystals (high-zinc and high-silica is the mechanism for crystalline glazes). G2931K on the right has much more boron, double the Al2O3, less SiO2 and is magnesia-alkali instead of zinc-alkali. It is the product of dozens of tests to find an ultra-clear having a glassy smooth surface. This particular chemistry, although having only a 6:1 SiO2:Al2O3 ratio is ultra-gloss. In addition, is has low expansion, will fast fire and the boron is not high enough to compromise the hardness.
Three low fire bodies need three different clear glazes. Why?

This picture has its own page with more detail, click here to see it.
Glaze fit. The left-most clay mug contains no talc (Plainsman Buffstone), the centre one about 25% talc (L212) and the right one is about 45% talc (L213). Talc raises thermal expansion. The centre glaze is G2931K, it is middle-of-the-road thermal expansion (Insight-live reports it as 7.4) and fits the low-talc bodies (and Zero3 porcelain and stoneware). But it crazes on Buffstone and shivers on L213. The lesson is: Forget about expecting one clear or base glaze to fit all low fire bodies. But there is a solution. I adjusted it to reduce its expansion to work on zero-talc porous bodies and raise it to work on high talc bodies. How? By decreasing and increasing the KNaO (in relation to other fluxes). The three fire crystal clear and work the best in a drop-and-hold firing.
Two low fire transparent highly fritted glaze recipes for pottery

This picture has its own page with more detail, click here to see it.
These work well from cone 04 up, they are better than any commercial transparent brushing glaze we have used. And far better than glazes made using raw sources of boron (like ulexite, colemanite, Gerstley Borate). These glazes have lower thermal expansion and do not craze on any body we have tried (yet are ultra gloss and ultra-clear). They are G1916QL1 and G3879C. We developed them for use on the dolomite-based (rather than talc-based) L4410L low-temperature art clay body. These are a good demonstration of the technical and economic sense it makes to use highly fritted glazes at low temperatures. Having a good base glaze is the key to adopting low temperatures for your production. You would likely agree that no stoneware glaze has melting patterns like these shown in this melt fluidity test! These recipes and all details about their development and adjustment are openly available.
These two transparent glazes are opposites:
In melt fluidity and surface tension

This picture has its own page with more detail, click here to see it.
This cone 04 flow tester compares two commercial low-fire transparent glazes. Their different chemistry strategies are revealed by the shape of these melt flows. While 3825B appears to have the higher melt fluidity, it also has much higher melt surface tension. This is evident in the narrow, rope-like stream and the way the flow meets the runway at a high angle before pulling into a rounded bead. A, by contrast, spreads and wets the runway, meandering downward in a broad, flat and relatively bubble-free river.
This difference is important in low-fire ware because these glazes must pass far more gases and bubbles than high-temperature glazes. The lower surface tension of A aids bubble release and healing after bubbles break. A is Amaco LG-10. B is Crysanthos SG213 (Spectrum 700 behaves similarly, although flowing less). Both approaches have advantages and disadvantages and are worth testing in your application.
Boron blue in low fire transparent glazes

This picture has its own page with more detail, click here to see it.
This high-boron high-CaOcone 04 glaze is generating calcium-borate crystals during cool down (called boron-blue). This is a common problem and a reason to control the boron levels in transparent glazes; use just enough to melt it well. If more melt fluidity is needed, decrease the percentage of CaO in favor of a lower melting oxide, that will certainly help. There is a positive: For opaque glazes, this effect can actually enable the use of less opacifier.
Bubbles in Terra Cotta transparent glazes. What to do?

This picture has its own page with more detail, click here to see it.
Two transparent glazes applied thickly and fired to cone 03 on a terra cotta body. Right: A commercial bottled clear, I had to paint it on in layers, I ended up getting it on pretty thick. Left: G1916S, a mix of Ferro frits, nepheline syenite and kaolin - one dip for 2 seconds and it was glazed. And it went on more evenly. Bubbles are, of course, generated by and clay body during firing, but terra cottas are the worst. And when fired toward vitrification the gas volume can really increase. Complicating this is the fact that low temperature glazes melt early, while body gassing may still be happening. Improvements? Both of these could have been applied thinner. And I could have fired them using a drop-and-hold and a slow-cool schedule. But the biggest improvement would likely be firing lower, to cone 04.
G1947U cone 10 transparent on Plainsman H550 and H570

This picture has its own page with more detail, click here to see it.
This is a base recipe that was originally used for electrical insulators on a 25% porcelain recipe. Since most porcelains and whitewares used in high fire ceramics have this same type of formulation, this glaze recipe has proven to work well. It is not highly fluid, so if refractory colorants are added extra flux may be needed.
What material makes the tiny bubbles? The big bubbles?

This picture has its own page with more detail, click here to see it.
These are two 10 gram GBMF test balls of Worthington Clear glaze fired at cone 03 on terra cotta tiles (55 Gerstley Borate, 30 kaolin, 20 silica). On the left it contains raw kaolin, on the right calcined kaolin. The clouds of finer bubbles (on the left) are gone from the glaze on the right. That means the kaolin is generating them and the Gerstley Borate the larger bubbles. These are a bane of the terra cotta process. One secret of getting more transparent glazes is to fire to temperature and soak only long enough to even out the temperature, then drop 100F and soak there (I hold it half an hour).
Devitrification of a transparent glaze

This picture has its own page with more detail, click here to see it.
This glaze consists of micro fine silica, calcined EP kaolin, Ferro Frit 3249 MgO frit, and Ferro Frit 3134. It has been ball milled for 1, 3, and 6 hours with these same results. Notice the crystallization that is occurring. This is likely a product of the MgO in the Frit 3249. This high boron frit introduces it in a far more mobile and fluid state than would talc or dolomite and MgO is a matting agent (by virtue of the micro crystallization it can produce). The fluid melt and the fine silica further enhance the effect.
How to reverse-engineer a commercial transparent glaze

This picture has its own page with more detail, click here to see it.
The commercial cone 04 clear brushing glaze (on the left) works really well on our clay bodies so I sent it away to be analyzed (about $130). That revealed high Al2O3/SiO2 levels, this explains its resistance to crazing on our clay bodies and, even better, indicates high durability. In my account at insight-live.com I was able source the same chemistry from two Fusion frits (plus a little kaolin and silica). The melt fluidities are almost identical (my G3879 has a little more surface tension). I needed to make a dipping glaze version and chose a method that would produce a thixotropic slurry. One caution: An assay lab cannot analyze the complexities of a colored glaze, instead focus on the base clear and add stains to that. The first two-gallon bucket made saved the development cost plus more! And knowing the recipe made it possible to adjust for even lower thermal expansion. Another plus: I can now make my own low SG or high SG brushing version.
These Stoneware Potters Do Something Unusual:
They make certain pieces using the low fire process

This picture has its own page with more detail, click here to see it.
Come 6 stoneware is super for most pieces. But there is some things you just cannot do. For example, you can decorate the underside! The one on the right is the back side of the plate. This is Plainsman Snow clay, it has 25% absorption at cone 06-04, this level of porosity is so high that the body can actually absorb the molten glaze if it is melt-fluid enough. However, when fired at cone 06 the body does not absorb any of the glaze. And the plates stay flat when fired on stilts. These are done by the team of Micah & Jeremiah Wassink of Creston, BC (at Pridham Studio). They make matching mugs, but fire those at cone 6 using underglaze decoration with a clear overglaze. But these plates are decorated using a combination of heavily pigmented viscous-melt low-fire glazes and a black underglaze and then finished with a thin layer of transparent glaze. Since the glaze is not crazed, water is not able to penetrate into the body. And the layer of clear glaze isolates the heavily pigmented colors from food. Even though they are low fire, these pieces are functional and food safe.
Inbound Photo Links
Links
| Glossary |
Colorant
In ceramics and pottery, colorants are added to glazes as metal oxides, metal-oxide-containing raw materials or as manufactured stains. |
| Glossary |
Opacifier
Glaze opacity refers to the degree to which it is opaque. Opacifiers are powders added to transparent ceramic glazes to make them opaque. |
| Glossary |
Boron Blue
Boron blue is a glaze fault involving the crystallization of calcium borate. It can be solved using glaze chemistry. |
| Glossary |
Crystallization
Ceramic glazes form crystals on cooling if the chemistry is right and the rate of cool is slow enough to permit molecular movement to the preferred orientation. |
| Glossary |
Glaze Durability
Ceramic glazes vary widely in their resistance to wear and leaching by acids and bases. The principle factors that determine durability are the glaze chemistry and firing temperature. |
| Glossary |
Liner Glaze
Liner-glazing is a way to assure that your ware has a durable and leach resistant surface. It also signals to customers that you care about this. |
| Glossary |
Glaze Bubbles
Suspended micro-bubbles in ceramic glazes affect their transparency and depth. Sometimes they add to to aesthetics. Often not. What causes them and what to do to remove them. |
| Glossary |
Translucency
A highly sought-after property in porcelain, fired close enough to melting to take on the glass-like property of passing light. Translucency implies tendency to warp during firing. |
| Glossary |
Ceramic Glaze
Ceramic glazes are glasses that have been adjusted to work on and with the clay body they are applied to. |
| Glossary |
Underglaze
Understand pottery underglazes: Why they brush differently, how they fire, why clears fail over them, and how to make your own recipes. In technical rather than art language. |
| Articles |
Concentrate on One Good Glaze
It is better to understand and have control of one good base glaze than be at the mercy of dozens of imported recipes that do not work. There is a lot more to being a good glaze than fired appearance. |
| Articles |
High Gloss Glazes
A transcript of a presentation at the 3rd Whitewares conference at Alfred University in the spring of 2000 by Richard Eppler. |
| Properties | Glaze Color |
| Recipes |
G1947U - Cone 10 Glossy transparent glaze
Reliable widely used glaze for cone 10 porcelains and whitewares. The original recipe was developed from a glaze used for porcelain insulators. |
| Recipes |
G2926B - Cone 6 Whiteware/Porcelain transparent glaze
A base transparent glaze recipe created by Tony Hansen, it fires high gloss and ultra clear with low melt mobility. |
| Recipes |
G1916Q - Low Fire Highly-Expansion-Adjustable Transparent
An expansion-adjustable cone 04 transparent glaze made using three common Ferro frits (low and high expansion), it produces an easy-to-use slurry. |
| Recipes |
G2931K - Low Fire Fritted Zero3 Transparent Glaze
A cone 03-02 clear medium-expansio glaze developed from Worthington Clear. |
Video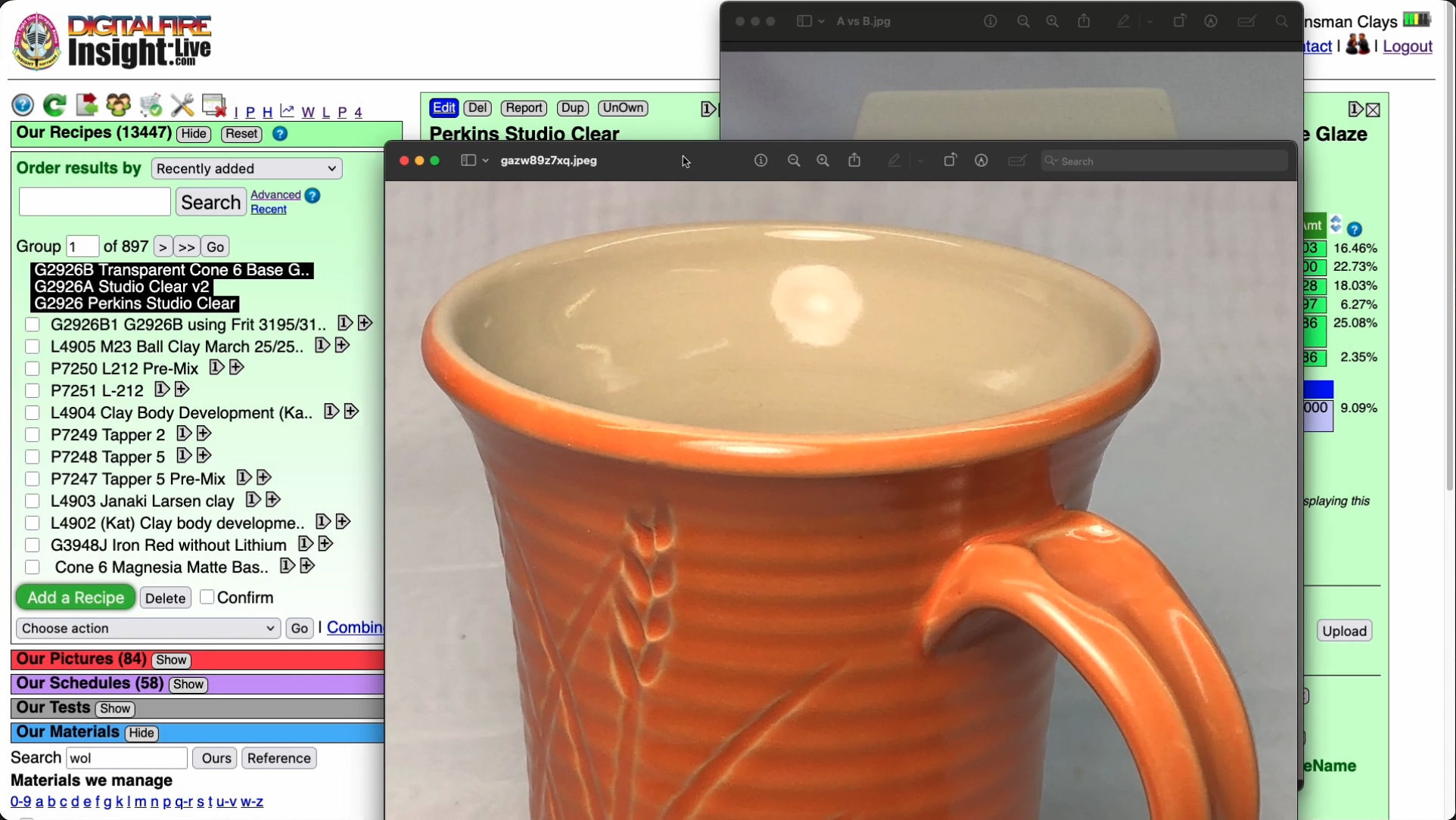 |
How I Developed the G2926B Cone 6 Transparent Base Glaze
How I found a pottery glaze recipe on Facebook, substituted a frit for the Gerstley Borate (using glaze chemistry), compared using a melt flow tester, added as much extra SiO2 as it would tolerate, and got a durable and easy-to-use cone 6 clear. |
| Troubles |
Clouding in Ceramic Glazes
There a many factors to deal with in your ceramic process to achieve transparent glazes that actually fire to a crystal-clear glass |
 PayPal | No tracking, No ads, No paywall, No transient content! Just organized, concise information constantly updated and improved. Was this helpful? Consider supporting me. |
| By Tony Hansen Follow me on        |  |
Got a Question?
Buy me a coffee and we can talk

https://digitalfire.com, All Rights Reserved
Privacy Policy





