Ferro Frit 3124
Alternate Names: F3124
Description: Leadless high calcium borosilicate frit
| Oxide | Analysis | Formula | Tolerance |
|---|---|---|---|
| CaO | 14.28% | 0.70 | |
| Na2O | 6.40% | 0.28 | |
| Al2O3 | 10.01% | 0.27 | |
| SiO2 | 54.94% | 2.50 | |
| K2O | 0.68% | 0.02 | |
| B2O3 | 13.74% | 0.54 | |
| Oxide Weight | 274.12 | ||
| Formula Weight | 274.12 | ||
Notes
This is a USA pottery frit. Ferro now calls it Frit 3124-2.
This borosilicate frit is high in calcium. It melts are very low temperatures and among the most useful of all common frits because of its glaze-like balanced chemistry. This frit has a chemistry somewhat similar to 3134 (the latter adds CaO, Na2O and B2O3 at the expense of all the Al2O3 and some SiO2.
Its stated intention is a calcium boron source for partially fritted glazes for wall tile and pottery, also in lead bisilicate dinnerware glazes in the cone 3-5 range. However, within pottery circles, like frit 3195 this frit is almost a complete glaze at low temperatures (requiring only a 10-20% addition of kaolin to suspend it). It has a medium thermal expansion and fits most bodies. However if glazes shiver some of this can be traded for Frit 3110. If they craze some can be substituted for Frit 3249. Frit 3124 is often added to glazes to make them melt lower, this works well because it is quite balanced already as a glaze, the net effect of adding it is to increase the boron content without overly disrupting the balance of other oxides.
Since the chemistry is high in CaO, it will affect browns and iron oxide colors.
Related Information
These common Ferro frits have distinct uses in traditional ceramics

This picture has its own page with more detail, click here to see it.
I used Veegum to form 10 gram GBMF test balls and fired them at cone 08 (1700F). Frits melt really well, they do have an LOI like raw materials. These contain boron (B2O3), it is a low expansion super-melter that raw materials don’t have. Frit 3124 (glossy) and 3195 (silky matte) are balanced-chemistry bases (just add 10-15% kaolin for a cone 04 glaze, or more silica+kaolin to go higher). Consider Frit 3110 a man-made low-Al2O3 super feldspar. Its high-sodium makes it high thermal expansion. It works really well in bodies and is great to make glazes that craze. The high-MgO Frit 3249 (made for the abrasives industry) has a very-low expansion, it is great for fixing crazing glazes. Frit 3134 is similar to 3124 but without Al2O3. Use it where the glaze does not need more Al2O3 (e.g. already has enough clay). It is no accident that these are used by potters in North America, they complement each other well (equivalents are made around the world by others). The Gerstley Borate is a natural source of boron (with issues frits do not have).
Melting range is mainly about boron content

This picture has its own page with more detail, click here to see it.
Fired at 1850. Notice that Frit 3195 is melting earlier. By 1950F, they appear much more similar. Melting earlier can be a disadvantage, it means that gases still escaping as materials in the body and glaze decompose get trapped in the glass matrix. But if the glaze melts later, these have more time to burn away. Glazes that have a lower B2O3 content will melt later, frit 3195 has 23% while Frit 3124 only has 14%).
Melt fluidity comparison - 1750F

This picture has its own page with more detail, click here to see it.
Fired at 350F/hr to 1750F and held for 15 minutes. Frit 3110 has taken off. And F75, 3195 and 3134 (the latter two having big differences in surface tension).
Various frits fired at 1950F

This picture has its own page with more detail, click here to see it.
These sixteen GBMF test balls have melted down onto a slab of grogged clay. Kiln fired at 108F/hr for the last 100 degrees F and held for 15 minutes. This demonstrates the comparison value of this test and how various frits compare in their melting character.
Frits fired to 2050F

This picture has its own page with more detail, click here to see it.
These are higher temperature frits. 10 gram balls were melted on to this tile.
Various frits fired at 1850F

This picture has its own page with more detail, click here to see it.
16 GBMF tests on a slab of grogged clay. Kiln fired at 108F/hr for last 100 degrees F and held for 15 minutes.
LOI horse race with surprising winners
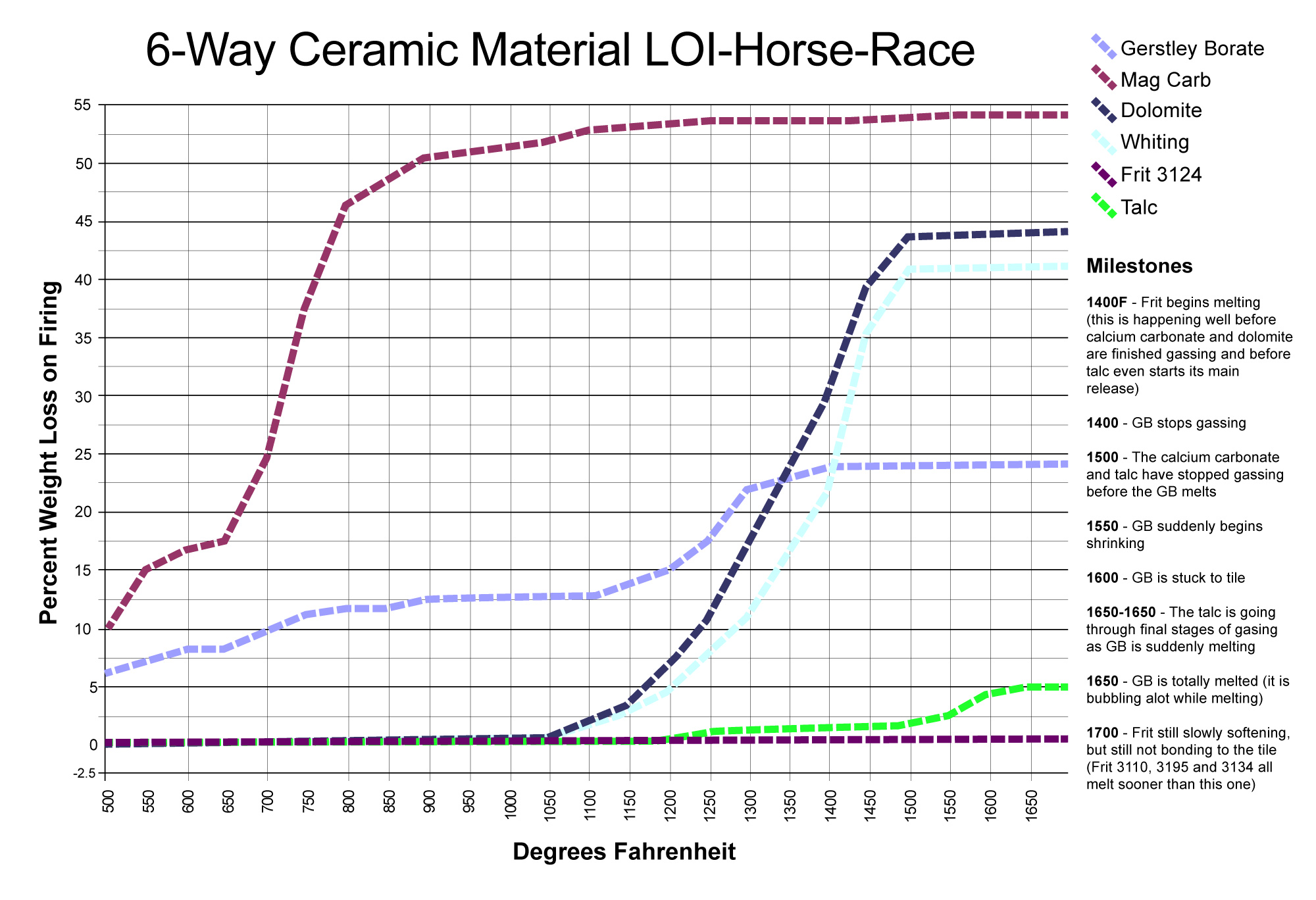
This picture has its own page with more detail, click here to see it.
This chart compares the decompositional off-gassing (% weight Loss on Ignition) behavior of six glaze materials as they are heated through the range 500-1700F. It is amazing how much weight some can lose on firing - for example, 100 grams of calcium carbonate generates 45 grams of CO2! This chart is a reminder that some late gassers overlap early melters. That is a problem. The LOI of these materials can affect glazes (causing bubbles, blisters, pinholes, crawling). Talc is an example: It is not finished gassing until 1650F, yet many fritted glazes have already begun melting by then. Even Gerstley Borate, a raw material, begins to melt while talc is barely finished gassing. Dolomite and calcium carbonate Other materials also create gases as they decompose during glaze melting (e.g. clays, carbonates, dioxides).
These two frits have one difference in the chemistry: Al2O3.

This picture has its own page with more detail, click here to see it.
These two boron frits (Ferro 3124 left, 3134 right) have almost the same chemistry. But there is one difference: The one on the right has no Al2O3, the one on the left has 10%. Alumina plays an important role (as an oxide that builds the glass) in stiffening the melt, giving it body and lowering its thermal expansion, you can see that in the way these flow when melting at 1800F. The frit on the right is invaluable where the glaze needs clay to suspend it (because the clay can supply the Al2O3). The frit on the left is better when the glaze already has plenty of clay, so it supplies the Al2O3. Of course, you need to be able to do the chemistry to figure out how to substitute these for each other because it involves changing the silica and kaolin amounts in the recipe also.
Five frits. One kiln at 1850F. Big chemistry drama.

This picture has its own page with more detail, click here to see it.
Five common North American Ferro Frits fired at 1850F on alumina tiles (each started as a 10-gram GBMF test ball and flattened during the firing). At this temperature, the differences are more evident than at 1950F. The degree of melting corresponds mainly, but not only, to the percentage of B2O3 present. Frit 3134 is the runaway leader because it couples that with almost no Al2O3 to stabilize the melt. Notice also the crack pattern, it is also very high in Na2O and thus has a high COE. However, why does Frit 3110 melt so well even though it contains almost no B2O3? Even higher Na2O, it is a powerful flux. Its COE is even higher than Frit 3134, but it is thick enough to have resisted cracking far (but it will in time).
High B2O3 imparts better melt fluidity, but also fewer micro-bubbles

This picture has its own page with more detail, click here to see it.
A cone 6 firing. The glaze on the left has a B2O3 molar content of 0.54 whereas the one on the right has 0.64 (other oxide levels are the same). This is triple the typical amount of boron in a cone 6 glaze, the result is obvious: High melt fluidity for both. But G3904A has a significant difference: The flow is more transparent because of the lower micro-bubble population. It's melt better enables the bubbles to pass, exit and the surface to heal. Why don't all glazes use more boron? Cost. Frits are expensive and they are the best source of boron. There is also a cost to durability (although mitigated when there is plenty of Al2O3 and SiO2 present, as is the case here). These recipes were part of a project to fix a recipe where the potter mistakenly used Frit 3134 instead of 3124 when mixing a large batch of glaze. I calculated how much kaolin and silica to add to bring the chemistry back into line with the original. This was possible because frit 3134 chemistry is an approximate oxide-subset of 3124. The resultant glaze is potentially better than the original.
1700F Frit Melt-Off: Who is the winner? Not the lead bisilicate!

This picture has its own page with more detail, click here to see it.
These were 10g balls melted using our GBMF test. We fired at a temperature far lower than typical bisque, notice how many of them are already melting well! Frit 3602 is lead bisilicate. But it got "smoked" by the Fusion FZ-16 high-zinc, high-boron zero-alumina! Maybe you always thought lead was the best melter. That it produced the most transparent, crystal-clear glass. But that is not what we see here. That being said, notice the lead is not crazing but the FZ-16 is crazing badly, that is a problem for many applications using this frit, it relies on a high percentage of KNaO. Notice something else: Each frit has a distinctive melt fingerprint that makes it recognizable in tests like this. Want to get some of this frit for pottery? You can't, Fusion Ceramics doesn't want to handle retail sales of smaller quantities.
Frit melt fluidity comparison - 1300F

This picture has its own page with more detail, click here to see it.
Fired at 350F/hr to 1300F and held for 15 minutes. Some are still burning off carbon (which seems strange). There are two early leaders: Ferro frit 3110 and Fusion frit F75 are starting to deform (they have almost the same chemistry). Amazingly, these two frits have low boron, they rely on high soda as the flux.
Melt fluidity comparison of frits - 1350F

This picture has its own page with more detail, click here to see it.
Fired at 350F/hr to 1350F and held for 15 minutes. Some are still burning off carbon (which seems strange). The two FZ16s are starting to move. Frit 3134 is expanding. 3602 is also starting to melt.
Melt fluidity comparison of frits - 1400F

This picture has its own page with more detail, click here to see it.
Fired at 350F/hr to 1400F and held for 15 minutes. Frit 3134 is still expanding. 3602 is also starting to flow. A number of them are shrinking and densifying like a porcelain would.
Frit Melt Fluidity Comparison - 1800F

This picture has its own page with more detail, click here to see it.
Fired at 350F/hr to 1800F and held for 15 minutes (I already did firings from 1300F-1750F in 50 degree increments, all of them are visible in the parent project). Frit 3110, 3134, 3195, F75 have run all the way down. All of the frits have softened and melted slowly over a range of temperatures (hundreds of degrees). By contrast, Gerstley Borate, the only raw material here, suddenly melted and flowed right over the cliff (between 1600 and1650)! But not before Frit 3602 and FZ16 had done so earlier. Frit 3249 is just starting to soften but F69 (the Fusion Frits equivalent) is a little ahead of it. LA300 and Frit 3124 are starting also. F524, F38, F15 will all be over the end by the next firing. The melt surface tension is evident by the way in which the melts spread out or hold together.
Melt fluidity comparison of frits - 1450F

This picture has its own page with more detail, click here to see it.
Fired at 350F/hr to 1450F and held for 15 minutes. Frit 3134 is still expanding. 3602 is blasting out of the gate, taking the lead. F75 is starting to flow.
Melt fluidity comparison of frits - 1500F

This picture has its own page with more detail, click here to see it.
Fired at 350F/hr to 1500F and held for 15 minutes. Frit 3134 is still expanding. 3602 and FZ16 are really starting to move. 3195, F38 and F15 are softening.
Melt fluidity comparison of frits - 1550F

This picture has its own page with more detail, click here to see it.
Fired at 350F/hr to 1550F and held for 15 minutes. Frit 3134 is still expanding. 3602 and FZ16 are going to be off-ramp by next firing.
Melt fluidity comparison of frits - 1650F

This picture has its own page with more detail, click here to see it.
Fired at 350F/hr to 1650F and held for 15 minutes. FZ16 has turned crystal clear and spread out across the runway (has low surface tension). Frit 3110 has so much surface tension that the flow can be lifted off the tester. Since 1600F Gerstley Borate has gone from unmelted to passing all the rest!
Melt fluidity comparison of frits - 1700F

This picture has its own page with more detail, click here to see it.
Fired at 350F/hr to 1700F and held for 15 minutes. 3110 is finally starting to move. 3134 also (being full of bubbles). Gerstley Borate has turned almost transparent (because the Colemanite portion of it is now melting). 3195 is looking very well behaved compared to most others, forming a bubble free glass of high surface tension (F15 and F524 are starting to do the same).
Substitute Ferro Frit 3134, using glaze chemistry, in three glaze types

This picture has its own page with more detail, click here to see it.
Can't get frit 3134 for glaze recipes? Can you replace it with frit 3124? No, 3124 has five times the amount of Al2O3 (the second most important oxide in glazes) and half the amount of B2O3 (the main melter). This ten-minute video presents a glaze chemistry approach that is easier to do than you probably think. It deals with three different glaze recipe types lacking sufficient clay to suspend the slurry. Learn to source the needed oxides from two other Ferro frits, 3110 (or Fusion F-75) and 3195 (Fusion F-2), and end up with at least 15% kaolin in each. A unique approach is required in each situation. Two of the calculations produce improved slurry properties and one yields a recipe of significantly lower cost. If you have a recipe that needs this and need help please contact us.
Ferro Frit 3124 vs Fusion F-19 at cone 04

This picture has its own page with more detail, click here to see it.
On paper, Fusion Frit F-19 has a very similar chemistry to 3124. However, as can be seen here, it is flowing a little more and appears to have a lower surface tension. The entrained bubbles are bigger so the glass is more transparent. This brings up some of the same puzzling questions as did our tests on their Fusion Frit F621/19.
G2934 using Fusion Frit F-19 instead of Ferro 3124

This picture has its own page with more detail, click here to see it.
G2934 is a popular recipe and there has been alarm recently because of the difficulty in getting the Ferro frit and the variation in its quality in recent years. This motivated us to get a supply of the Fusion equivalent, F-19. When doing substitutions like this we do testing in glazes and with melt fluidity tests - like this GLFL test.
Ferro Frit 3124 vs Fusion F621/19 at cone 04

This picture has its own page with more detail, click here to see it.
Fusion Frit F621/19 is recommended as a substitute candidate for Ferro Frit 3124. However, as shown on this melt fluidity test of the two pure frit powders, it appears to have a lower surface tension and flow better. However the character of the flow is opposite to what is expected of a lower surface tension in the melt. There should either be a lower population of micro-bubbles or they should be smaller - but the opposite is the case. There should be fewer breaking at the surface and they should be healing better - but that is not the case. Frits are supposed to be free of carbon or hydrates - or are they? If the frit is sourcing gases because of raw materials not fully decomposing during smelting - how is that even possible? Do you know why this is happening, can you tell me please? Fusion Ceramics does not supply the chemistry of Frit F621/19 and it is not shown on their website in 2021, but I have no reason to believe it contains fluorine.
Frit Melt Fluidity Comparison - 1850F

This picture has its own page with more detail, click here to see it.
These melt flow tests were fired at 350F/hr to 1850F and held for 15 minutes (I did firings at 50-degree increments across a wide range). It is amazing how active some frits are, even well below normal bisque temperatures! Frit 3110, Frit 3134, Frit 3195, Frit F-75 have all flowed all the way down for many previous temperatures. LA300 and Frit 3124 were just starting at 1800F, look at them now! Frit F-524 and Frit F-38 have gone from half-way at 1800F to water-falling over the end. Frit 3249 is still not out-of-the-gate but Frit F-69 (the Fusion Frits equivalent of 3249) is half-way. Note how the melt surface tension is evident by the way in which the melts spread out or hold together. By contrast, Gerstley Borate (labelled "GB"), the only raw material here, suddenly melted and flowed right over-the-cliff between 1600 and 1650! The best melter of all of them is high-boron high-zinc Frit FZ-16.
Links
| Materials |
Ferro Frit 3134
A frit with 23% B2O3. The most common of frits used in pottery in North America. Around the world, other companies make frits of equivalent chemistry. |
| Materials |
General Frit GF-113
|
| Materials |
Fusion Frit F-19
A commonly available calcium borosilicate frit having a similar chemistry to Ferro Frit 3124. |
| Materials |
Hommel Frit 378A
|
| Materials |
Pemco Frit P-311
|
| Materials |
Hommel Frit 90
|
| Materials |
Frit RCG 2430
|
| Materials |
Ferro Frit 4124
|
| Materials |
PotteryCrafts Frit P3124
|
| Materials |
Potclays Frit 2272
|
| Materials |
Fusion Frit F621/19
A commonly available calcium borosilicate frit having a similar chemistry to Ferro Frit 3124. |
| Typecodes |
Frit
A frit is the powdered form a man-made glass. Frits are premelted, then ground to a glass. They have tightly controlled chemistries, they are available for glazes of all types. |
| URLs |
https://digitalfire.com/4sight/datasheets/ferropotteryfrits2008.pdf
Ferro Pottery Frits 2008 |
Data
| Co-efficient of Linear Expansion | 7.94 |
|---|---|
| Frit Melting Range (C) | 1600-1750F |
 PayPal | No tracking, No ads, No paywall, No transient content! Just organized, concise information constantly updated and improved. Was this helpful? Consider supporting me. |
| By Tony Hansen Follow me on        |  |
Got a Question?
Buy me a coffee and we can talk

https://digitalfire.com, All Rights Reserved
Privacy Policy
