Contact Me
Let's have a  together and answer your questions. together and answer your questions.
Other ways to Support My Work
Subscribe to Insight-Live.com. It is about DIY testing and development, not letting information slip away. Help Me on Social

Login to your online account
Chemistry plus physics.
Maintain your recipes, test results, firing schedules, pictures, materials, projects, etc.
Access your data from any connected device. Import desktop Insight data (and of other products).
Group accounts for industry and education. Private accounts for potters.
Get started.

Download for Mac, PC, Linux
Interactive glaze chemistry for the desktop. Free (no longer in development but still maintained, M1 Mac version now available).
Download here or in the Files panel within your Insight-live.com account.
What people have said about Digitalfire- Your resources are truly amazing and as an ex electronic engineer (now a potter), I really am impressed with your analytical approaches. Your site is almost a complete college level course on pottery (less the throwing & handbuilding). Thank you for your wonderful contributions.
- It would be indeed an honour for me if my contribution in any form can be of any use.
- Thanks a lot for this sea of knowledge.
- I found your site and spent a long time studying various parts of it. I intend to use it alot, and hence I probably purchase the software in due course.
- I'm half way through MF and have loads of questions but will wait untill I have finished and tried some experiments before I ask.
- Just wanted to tell you I just found your site and find it extremely useful. Got it
bookmarked and will be referring to it frequently. Great job! Thank you!
- Very useful articles on practical solutions to the every day problems Ceramic Technicians face in their organizations.
- Everything you guys post on this page is materials science nerd gold.
- Your site is excellent and informative, you should conduct online conferences on various subjects, please let me know if you have one.
- I am at the point where I'm ready to go beyond the hit or miss approach to finding workable glazes. So far I'm finding you book most helpful. It is clearly written, and
most of all understandable for a non-chemist like myself. I look forward to trying the Insight program soon. Your website is a great source of information. Thanks and keep up the great work.
What people have said about Insight-Live- I love your software and I really appreciate you being there when I need help. Keep on being amazing.
I am still enjoying insight every day. You were right - having it in the cloud is a huge advantage. Your service to potters worldwide is extraordinary. It strikes me as a remarkably generous and altruistic project, and the value it brings to our community is immense.
- Many thanks for your time and trouble, re the information cone 6. You are a wealth of generosity and help to the home Potter.
- I have done production pottery at cone 10 and 5 for 40 years, but during almost of full year of downtime due to COVID, I am exploring low fire terra cotta. Digital fire is an amazing resource. Thank you so much.
- Your site, I think, is the best source of information on ceramics that I have found. I have always wanted to know how things work rather than just doing it because someone said so. Your site gives reasons why and how to apply what you have learned! I've learned much from you and now I am enrolled in chemistry classes to better understand what you present on your site. Thank your for the Digital Fire Website and the Insight-live
Site.
- The knowledge and information you share on digital fire is a rare gem on the internet. I greatly appreciate your writing style. To the point and full of facts. I am wanting to be more active in my glaze creation and begin to make my own glazes. This, to me seems like a huge step away from the safe and what I know of the glazes I have been working with.
- And I want to add - you are doing a magnificent job with your webpage. Just unbelievable amount of work and information you put there. I have learned a lot from your webpage. Thank you.
- Please continue this brilliant service!
- After more than 50 years making pots, selling and teaching I’m done. Worn to a frazzle. Just wanted to let you know that over the years I’ve relied on you for information on materials and glazes. I’ve never been disappointed, and I owe you a big thanks. I’m over 80 and I need to slow down. I’ll continue making pottery, but not so much. I’ve been at it since 1968 (phew!). I thank you for your advice and especially for your website and its cascading information. Always useful, always on target. I’m only sad that I was not able to meet you in person. Peace, love and happiness.
- Thank you for all of the information you publish on your website. Digitalfire has been my primary resource for researching raw materials and I will be soon be subscribing to Insight. Thank you so much for all of your work. My fellow students and I refer to your website almost daily.
- I am writing to express my sincere gratitude for the incredible work you have done over the decades. A large part of my own journey and understanding as a ceramicist is built upon the knowledge I was able to learn through your work at Digitalfire and Insight-live. The value of this technical foundation is truly immeasurable, and I wanted to let you know how much it has meant to my development in this craft. Thank you for everything you have contributed to the joy and science of ceramics.
| June 2026: We are continuing a major code rewrite. Please contact us if you find issues. Thank you. I will send practical posts like these (from thousands I maintain). No ads or tracking. The first email will provide one-click unsubscribe. Signup is being email-bombed by bots. For now, please subscribe inside your insight-live.com account.
Blog
Same body, same outside glaze.
But the inside transparent glaze is different

Glazing black clay bodies stained with manganese is just about impossible with typical transparent glazes. The glaze over-fluxes the clay surface and ruins the color. Worse, if it accelerates surface maturity, the body can blister or generate LOI gases that blister the glaze. How about transparent glazes over a black engobe instead? At least the body color is not lost. But the wrong transparent glaze can do what you see here (inside left).
These mugs are a buff stoneware, Plainsman M340. A black engobe was applied by pouring the inside and dipping the outside two-thirds of the way down.
Left: A L3954F black engobe was applied inside and upper exterior at leather hard. After firing to cone 6 using the PLC6DS schedule, G2926B—which is crystal clear on M340 itself—became completely clouded over the engobe because bubbles generated during firing remained trapped in the melt.
Right: The entire mug was dipped in GA6-B. The Alberta Slip particles and the melt characteristics of GA6-B promote bubble coalescence and escape, producing an exceptionally glossy jet-black surface over the same engobe.
Monday 8th June 2026
No glaze chemistry needed
At least not right away

You have 147 glaze recipes. How can you get your head around all of them? Is glaze chemistry needed? No, that's a "maybe" way in the future. Right now, you need to start organized documentation. The recipe for each. A few pictures of each fired on different clay bodies, different thicknesses. Perhaps slow and fast-cooled firing. This is what an account at Insight-live does well. What it does even better is tracking your testing. The first step is to assign each recipe a proper code number (replacing these) and write that on all test specimens and buckets. From this point on, learn. Record every observation you make about each in its notes.
Through all of this, constant use in the studio (or factory) will never stop surfacing problems (e.g. settling in the bucket, crazing, running, blistering, material issues, etc.). The seriousness of each will determine the level of attack. First, identify the mechanism of the desired fired result. If it is a base recipe plus additions of colorants, opacifiers or variegators, then check if the base of one of the other glazes has a similar surface texture and character. If so, then could the additives in the troublesome one be used with the better base? If not, then it's time to sanity check the recipe and bring out the heavy guns of at least looking at the chemistry. But in Insight-live, you only need to turn on the display of the unity formula (there is nothing else to do). Next, make sure each material in the recipe links to one in the material database (so the calculated formula is accurate). Then compare the calculated unity formula with a limit formula (often a simple sanity check, like with the recipe, quickly spots oxides that are in excess or are short.
Thursday 4th June 2026
A transparent glaze is going satin:
Is it the feldspar and kaolin substitutions?
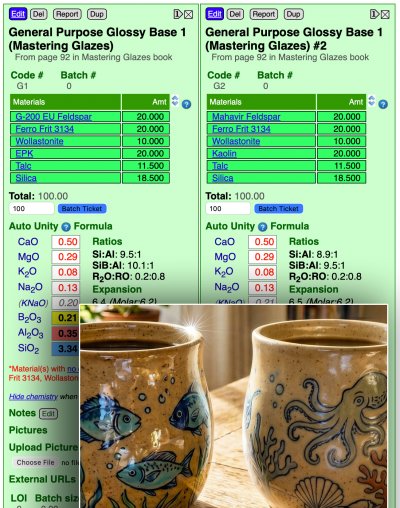
A potter reports that a switch from G-200 feldspar to Mahavir, and EPK to Imerys kaolin, has resulted in this transparent glaze becoming more satin. Is that possible? Yes. Because this glaze is on a unity formula tipping point.
To see it, you do not need to know how to do glaze chemistry, just how to display the calculated unity formulas side-by-side. My Insight-live shows them here. The material change has little effect. But there is an anomaly: 0.29 MgO. That is magnesia matte territory. The MgO is very likely there to help bring the thermal expansion as low as possible (to avoid crazing). For people who cool their kilns relatively quickly, this fires glossy. But a material change could well affect the cooling rate needed to maintain the gloss. That being said, the potter may also be firing slower, yet attributing the mattness to the materials. Or it could be a combination of both.
This is a popular glaze, among others in the book "Mastering Glazes". In Ron Roy's circumstances, and for many others, it is glossy. But for this potter, a small change (in the recipe materials and also likely in firing) has produced this issue.
Context: MGBase3, Tipping point
Thursday 4th June 2026
Alberta Slip as a functional honey-transparent base:
The glaze I reach for again and again
Are glazes food safe just because they carry a label?

This Gemini-generated mug could conceivably exist yet carry these labels. Yet experienced ceramic technicians would immediately be suspicious. The glaze is highly fluid and heavily crystallized; both suggest low or very low Al2O3 levels (it is the key oxide that makes glazes durable). If the interior color were produced using a cadmium-containing encapsulated stain, cadmium-release testing would be essential before claiming the ware is food safe. This is clearly engineered for visual effects rather than durability. None of those characteristics prove it is unsafe, but they do mean that labels like "nontoxic" are not substitutes for actual leach testing. A glaze can be made entirely from materials classified as nontoxic and still fail to meet the durability standards expected of functional foodware.
Context: Commercial glazes on decorative.., ASTM D-4236 - Standard..
Thursday 4th June 2026
Should I glaze the outside of this mug now? No!

This bisque mug has been glazed on the inside. But the bisque has absorbed water from that glaze, and this thin-walled mug is now waterlogged as a result (except at the thicker base). It does not have the absorbency needed to build up a thick enough layer of glaze on the outside. Even if it did, the water from the two glazes would wet the bisque so much that its drying time would be greatly extended. This is a problem because the mechanism of attachment of glaze to the body is fragile and works best when the glaze dries quickly. When drying is too very slow, bubbling and cracking often occur (leading to crawling in the firing).
Context: Does bisque ware need.., Glaze thickness
Monday 1st June 2026
Yixing teapot making. Is it magic?
Or highly evolved craft and science?

The Yixing teapot craftsmen appear to break all the rules and yet produce impossibly delicate and symmetrical pieces. Hao-Tong Yan, one of those craftsmen, and I have been trying to understand the technical reasons for how this amazing craft is possible. It turns out not to be magic, but actually a highly evolved understanding of a very unusual material. Here are some of the things that we are coming to understand (which is making it possible to create a facsimile of the clay in North America).
-The Yixing ore can have the appearance of being like rocks, yet they make a workable clay body from it.
-The clay appears highly plastic yet is not; the workability is coming from surprising places.
-The clay is stiff enough to resist deformation, yet is cohesive enough to join seamlessly.
-Craftsmen flatten the clay with a mallet, instead of rolling it, yet it does not stick to the board.
-Sections are simply glued with slip, yet they hold.
-The clay burnishes, yet is not smooth.
-Fired ware is smooth, yet the soft clay appears sandy.
-The fired surface is glossy, yet there is no glaze.
-The fired clay appears super dense yet does have porosity.
Context: Yixing Clay Teapots on.., Stunning video of Yixing..
Saturday 30th May 2026
2026: ChatGPT is doing credible troubleshooting

I uploaded a screenshot of this recipe panel from Insight-live and asked ChatGPT why sanitaryware using this glaze is dunting. Its response is impressive, good enough to provide remediation ideas.
-It notes things about the recipe that are unusual. For example, that the Al2O3 of 0.69 and Si:Al only 4:1, plus 13% calcined alumina, make this glaze more like a refractory enamel than a normal glaze.
-Its observation that crystallization could mean the real thermal expansion may be different from the calculated one, maybe much lower.
-Barium carbonate decomposition does not seem like something that would contribute to dunting, but its presence is strange for such a glaze.
Based on its answer, I think the 13% calcined alumina is the wild card, which is way too much for any glaze to dissolve, something to deal with first.
Context: ChatGPT was completely wrong.., ChatGPT is surprisingly wrong.., Two ChatBots square off.., Does ChatGPT know the.., Sanitary ware
Tuesday 26th May 2026
Crystalline glazes normally craze:
Here is one way to fix that

The mug on the left, made by Holly McKeen, is a typical cone 10 Grolleg kaolin mullite porcelain (highly vitrified, low in residual quartz). Its glaze is crazed. Crystalline glazes are high in Na2O, making crazing virtually certain. Since most pieces are decorative, crystal glazers just accept this as part of the process. But these are functional mugs, the glaze needs to fit (if only for ware strength).
But what if the thermal expansion of the body could be significantly raised? The body on the right is Crystal Ice, it contains 40% silica (vs 20-25% in a typical porcelain). The percentage of Nepheline has been reduced, lowering vitrification to about 1.5% porosity. As a result, more quartz survives undissolved and less mullite develops, raising the body’s thermal expansion. The result is a body with a much higher thermal expansion, so it can not only relieve the glaze tension but actually put a squeeze on it. There is a downside: These are less resistant to dunting and thermal shock failure during use.
Could the glaze be adjusted instead? Yes. Some of the Na2O could be substituted for Li2O, the latter is also a strong melter but has a much lower thermal expansion. Glaze chemistry could be used to source it from Spodumene (to avoid solubility issues with lithium carbonate). However, zinc-silicate crystalline glazes are very sensitive systems, so the more lithia is introduced the more likely the effect on the firing window, crystal size/density and background clarity.
Context: Crystalline glazes, Calculated Thermal Expansion
Tuesday 26th May 2026
Incorrect craze-fixing advice is still common online:
Well demonstrated using an AI-generated photo!

Crazing is one of the most common glaze defects. AI image generators can produce this really convincing photo, but AI explanations often still recycle oversimplified glaze-fit advice from the web. Let's work in reverse to see why using this speckled stoneware, it has lots of ball clay and quartz, it is easy to fit glazes to. What would it take to craze the glaze on the right? A lot. Glazes that craze out of the kiln on quartz-rich bodies are not "slightly misfit"; they are "hugely misfit". Under or over-firing, or holding less time at temperature, would not be enough to craze it. Reducing the silica enough to start severe crazing like this would fundamentally alter the glaze character and functionality.
This is not a recipe-level problem (e.g. reducing feldspar for silica or zinc). This is a material problem; it is an oxide chemistry issue. By far the best way to put the glaze under tension, to craze it, is to trade low thermal expansion oxides (not materials) for high ones. In this case, shift some of the flux unity away from CaO/MgO and toward KNaO (the latter being the highest thermal expansion oxides, by far).
All are fluxes and this is a transparent, so minimal change it character should occur.
Context: Glaze Crazing
Sunday 24th May 2026
|



















